QUESTION IMAGE
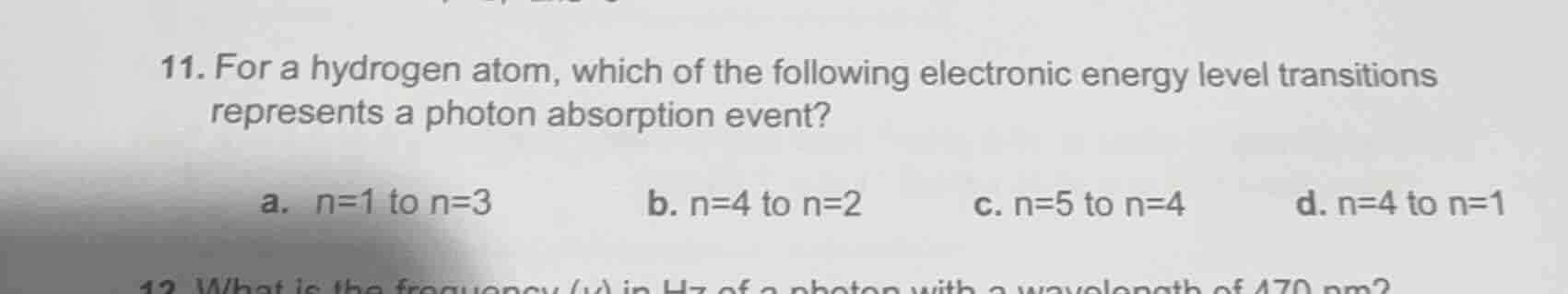
Question
- for a hydrogen atom, which of the following electronic energy level transitions represents a photon absorption event?
\ta. n=1 to n=3
\tb. n=4 to n=2
\tc. n=5 to n=4
\td. n=4 to n=1
12 what is the frequency (ν) in hz of a photon with a wavelength of 470 nm?
Brief Explanations
Photon absorption occurs when an electron in a hydrogen atom moves from a lower energy level (lower principal quantum number $n$) to a higher energy level (higher $n$). Emission happens when electrons move from higher to lower $n$.
- Option a: $n=1$ (lower) to $n=3$ (higher) is absorption.
- Options b, c, d: All involve electrons moving from higher to lower $n$, which are emission events.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
a. n=1 to n=3