QUESTION IMAGE
Question
3 of 14
actinide series**
89 ac (227)
90 th 232
91 pa 231
92 u 238
93 np (237)
94 pu (244)
95 am (243)
96 cm (247)
97 bk (247)
98 cf (251)
99 es (252)
100 fm (257)
101 md (258)
102 no (259)
103 lr (262)
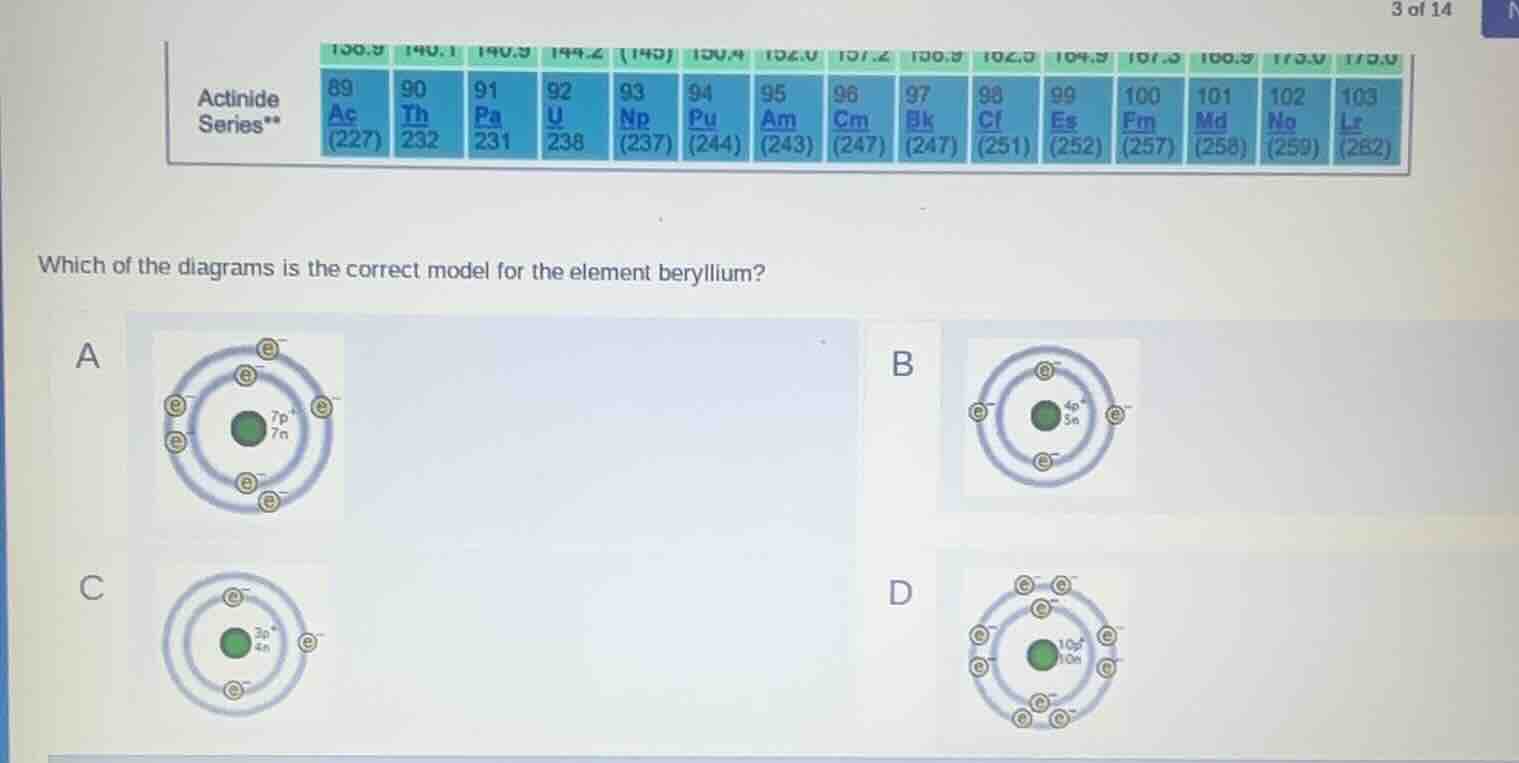
which of the diagrams is the correct model for the element beryllium?
a
b
c
d
Brief Explanations
- First, find beryllium's atomic number: it is 4. This means it has 4 protons ($p^+$) and, in a neutral atom, 4 electrons ($e^-$).
- Beryllium's most common isotope has 5 neutrons ($n$), so the nucleus has $4p^+ + 5n$.
- Use the electron shell rule: the first shell holds max 2 electrons, the remaining 2 electrons go to the second shell.
- Match this to the options: Option B has 4 protons, 5 neutrons, 2 electrons in the first shell, and 2 in the second shell.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
B. <Diagram with 4 protons, 5 neutrons, 2 inner-shell electrons, 2 outer-shell electrons>