QUESTION IMAGE
Question
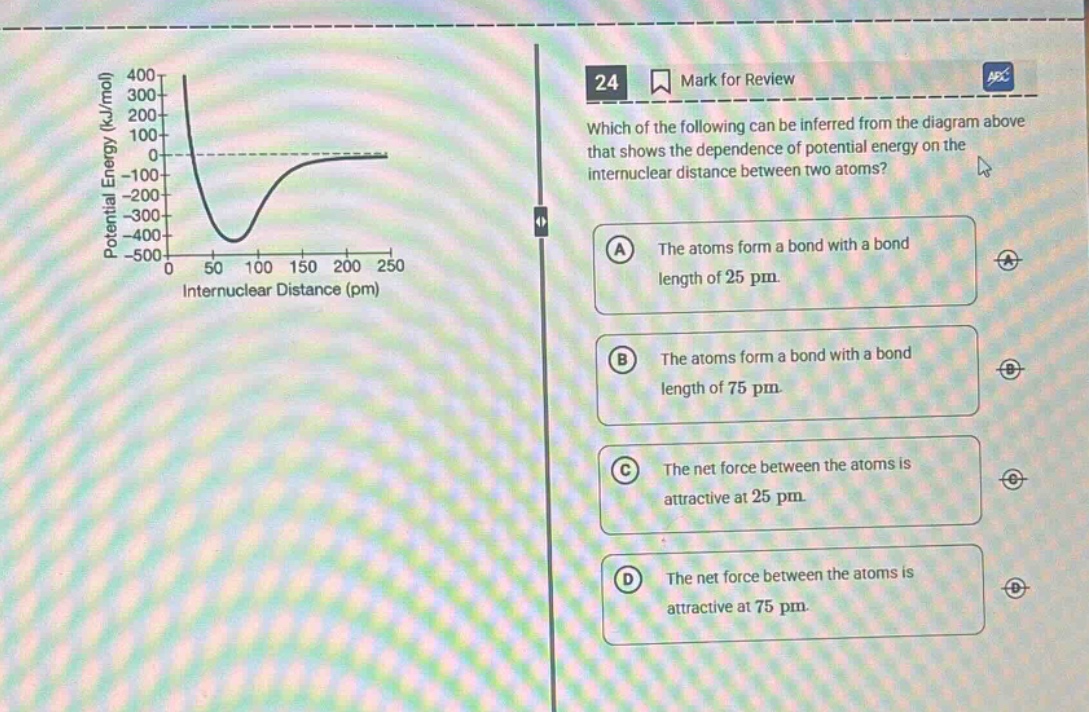
24 mark for review which of the following can be inferred from the diagram above that shows the dependence of potential energy on the internuclear distance between two atoms? a the atoms form a bond with a bond length of 25 pm. b the atoms form a bond with a bond length of 75 pm. c the net force between the atoms is attractive at 25 pm. d the net force between the atoms is attractive at 75 pm. (diagram: potential energy (kj/mol) on y - axis, internuclear distance (pm) on x - axis, curve showing potential energy vs internuclear distance)
- Analyze Bond Length: Bond length is the internuclear distance at the minimum potential energy (the lowest point of the curve). From the graph, the minimum is around 75 pm (not 25 pm), so A is wrong, B is a candidate.
- Analyze Force at Different Distances:
- At \( r < \text{bond length} \) (e.g., 25 pm, less than ~75 pm), the curve is decreasing (potential energy drops as \( r \) increases). The force is repulsive here (since pushing atoms closer than bond length requires energy, so they repel).
- At \( r = \text{bond length} \) (75 pm), force is zero (equilibrium).
- At \( r > \text{bond length} \) (e.g., 75 pm is the bond length, so distances just above? Wait, 75 pm is the bond length. Wait, the minimum is around 75 pm. Wait, when \( r = 75 \) pm (the bond length), force is zero. Wait, no—wait, the curve: as \( r \) increases from 0, potential energy decreases (repulsive force dominates, so as \( r \) increases, repulsion decreases, PE drops) until the minimum (bond length, where force is zero: attractive and repulsive forces balance). Then, as \( r \) increases beyond bond length, PE increases (attractive force dominates, so pulling atoms apart requires energy, PE rises). Wait, no—wait, the standard potential energy curve: at \( r < r_0 \) (bond length), PE decreases as \( r \) increases (repulsive force: \( F = -\frac{dU}{dr} \), so if \( U \) decreases with \( r \), \( \frac{dU}{dr} \) is negative, so \( F \) is positive (repulsive, pushing apart). At \( r > r_0 \), \( U \) increases with \( r \), so \( \frac{dU}{dr} \) is positive, \( F \) is negative (attractive, pulling together). At \( r = r_0 \), \( \frac{dU}{dr} = 0 \) (force zero).
So:
- At 25 pm (\( r < r_0 \)): \( U \) is decreasing as \( r \) increases (from 0 to 75 pm, PE drops). So \( \frac{dU}{dr} < 0 \), so \( F = -\frac{dU}{dr} > 0 \) (repulsive force). So option C (attractive at 25 pm) is wrong.
- At 75 pm: bond length (minimum PE, force zero). Wait, but option D says "the net force between the atoms is attractive at 75 pm"—no, at bond length, force is zero. Wait, maybe I misread the options. Wait, option D: "at 75 pm"—wait, the bond length is ~75 pm (the minimum). Wait, no—wait the graph: the minimum is around 75 pm (x-axis: 50, 100—wait, the x-axis is 0, 50, 100, 150... The minimum is between 50 and 100, maybe 75 pm. So bond length is ~75 pm (option B). Now, force at 75 pm: at bond length, force is zero (equilibrium). Wait, but option D: "attractive at 75 pm"—no, force is zero. Wait, maybe I messed up. Wait, let's re-express:
- Bond length is the internuclear distance at minimum potential energy (the lowest point of the curve). From the graph, the minimum is around 75 pm (between 50 and 100, closer to 75). So option B says bond length 75 pm: correct.
- For force:
- At 25 pm (less than bond length): the curve is decreasing (PE decreases as \( r \) increases). So \( \frac{dU}{dr} < 0 \), so \( F = -\frac{dU}{dr} > 0 \) (repulsive force). So option C (attractive at 25 pm) is wrong.
- At 75 pm: it's the bond length (minimum PE), so force is zero (balanced attractive and repulsive). So option D: "attractive at 75 pm" is wrong (force is zero).
So only option B is correct.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
B. The atoms form a bond with a bond length of 75 pm.