QUESTION IMAGE
Question
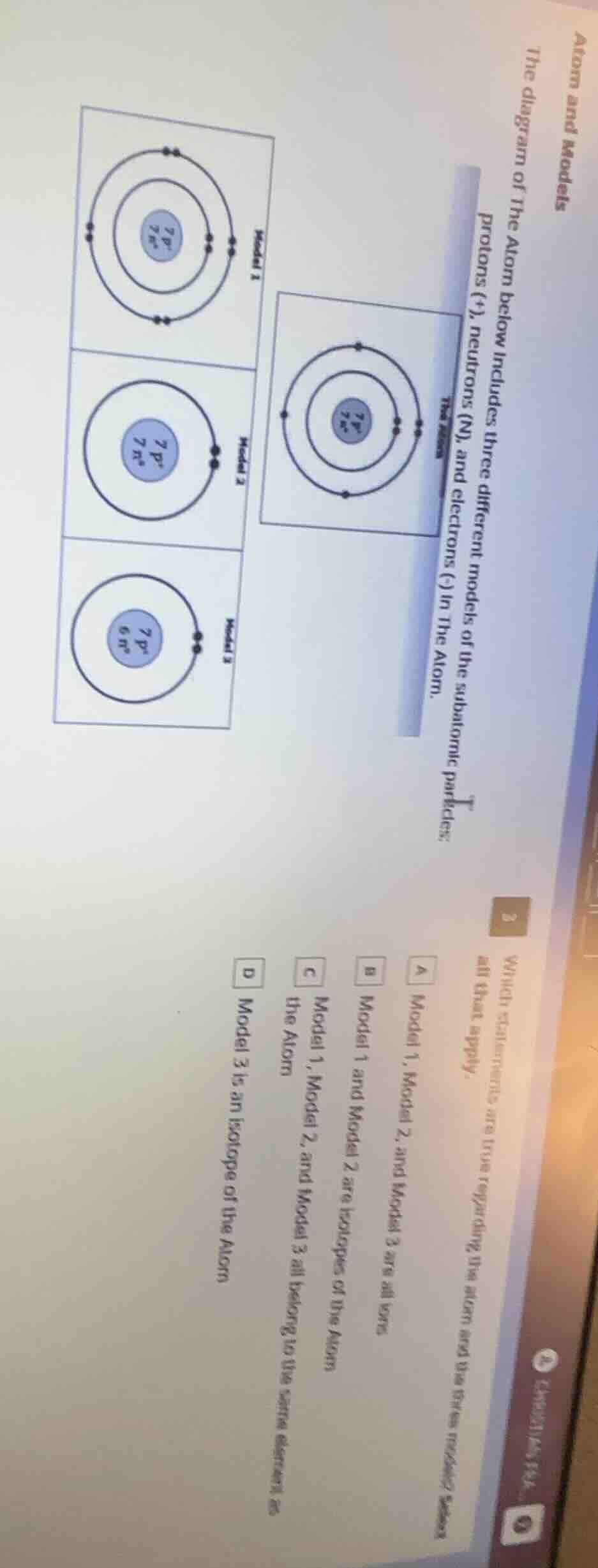
atom and models
the diagram of the atom below includes three different models of the subatomic particles: protons (+), neutrons (n), and electrons (-) in the atom.
which statements are true regarding the atom and its three models? select all that apply.
a model 1, model 2, and model 3 are all ions
b model 1 and model 2 are isotopes of the atom
c model 1, model 2, and model 3 all belong to the same element as the atom
d model 3 is an isotope of the atom
- First, recall the definition of an isotope: atoms with the same number of protons (same element) but different numbers of neutrons.
- For Model 1: Protons ($P$) = 7, Neutrons ($n$) = 7, Electrons = let's check (but isotope depends on $P$ and $n$).
- Model 2: $P = 7$, $n = 7$ (wait, no, wait the diagrams: Wait, Model 1: nucleus 7P,7n; Model 2: 7P,7n? Wait no, maybe I misread. Wait the problem: Model 1: nucleus 7P,7n; Model 2: 7P,7n? Wait no, Model 3: 7P,6n. Wait, no, let's re-express:
- Isotopes have same atomic number (same $P$) but different mass number (different $n$).
- Model 1: $P=7$, $n=7$
- Model 2: $P=7$, $n=7$ (wait, no, maybe Model 2's nucleus: 7P,7n? Wait the diagram: Model 1: two electron shells, Model 2: one shell? Wait no, maybe the neutrons: Model 1: 7n, Model 2:7n, Model 3:6n. Protons: all 7.
- So Model 1 and Model 2: same $P$ and same $n$ → same atom, not isotopes. Model 1 (7n) and Model 3 (6n): same $P$, different $n$ → isotopes. Model 2: 7n, same as Model 1. So:
- Option A: Model 1,2,3 all ions? Ions have different number of electrons than protons. Model 1: let's count electrons. Model 1: inner shell 2, outer shell 5 (total 7). Protons 7 → neutral. Model 2: outer shell 2? Wait no, Model 2: one shell with 2 electrons? Wait protons 7, electrons 2? No, that can't be. Wait maybe I misread the electron dots. Let's re-express:
- Wait the problem says "protons (+), neutrons (N), and electrons (-)". So for an atom to be neutral, electrons = protons.
- Model 1: Protons=7, Neutrons=7. Electrons: let's count the dots. Inner shell (first) can hold 2, outer (second) up to 8. Model 1: inner shell 2, outer shell 5 (2+5=7) → electrons=7, protons=7 → neutral.
- Model 2: Protons=7, Neutrons=7. Electrons: one shell with 2? No, that would be ion (7 protons, 2 electrons → charge +5). But maybe Model 2's electrons: two dots (electrons) → 2 electrons. So protons 7, electrons 2 → ion. Model 3: Protons=7, Neutrons=6. Electrons: two dots → 2 electrons? No, wait Model 3's nucleus: 7P,6n. Electrons: two dots → 2. So protons 7, electrons 2 → ion. Wait this is confusing. Maybe the key is isotopes: same $P$, different $n$.
- Model 1: $P=7$, $n=7$
- Model 2: $P=7$, $n=7$ (same as Model 1)
- Model 3: $P=7$, $n=6$ (different $n$ from Model 1 and 2)
- So isotopes: atoms with same $P$, different $n$. So Model 1 (7n) and Model 3 (6n) are isotopes. Model 2: same $n$ as Model 1 → same atom.
- Now let's check options:
- Option D: "Model 3 is an isotope of the Atom" → The atom (let's say the original is Model 1: 7P,7n). Model 3:7P,6n → same $P$, different $n$ → isotope. So D is correct.
- Option C: "Model 1, Model 2, and Model 3 all belong to the same element as the Atom" → Element is determined by protons (atomic number). All have 7 protons → same element (nitrogen, since atomic number 7 is N). So even if they are ions or isotopes, same element. So C is correct? Wait the "Atom" here: let's assume the Atom is the neutral one (Model 1: neutral, 7P,7n,7e). Model 2: 7P,7n,2e (ion) → same element (N). Model 3:7P,6n,2e (ion) → same element (N). So C: all belong to same element. Correct?
- Wait the options:
- A: Model 1,2,3 all ions? Model 1: neutral (e=7, p=7) → not ion. So A is wrong.
- B: Model 1 and Model 2 are isotopes? Model 1: n=7, Model 2: n=7 → same n → not isotopes. So B wrong.
- C: Model 1,2,3 all belong to same element (since same P=7) → correct.
- D: Model 3 is isotope of the Atom (Atom is Model 1: 7P,7n; Model 3:7P,6n → same P, different n → isotope) → correct.
- Wait but the options: Let's recheck the problem statement. T…
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
C. Model 1, Model 2, and Model 3 all belong to the same element as the Atom
D. Model 3 is an isotope of the Atom