QUESTION IMAGE
Question
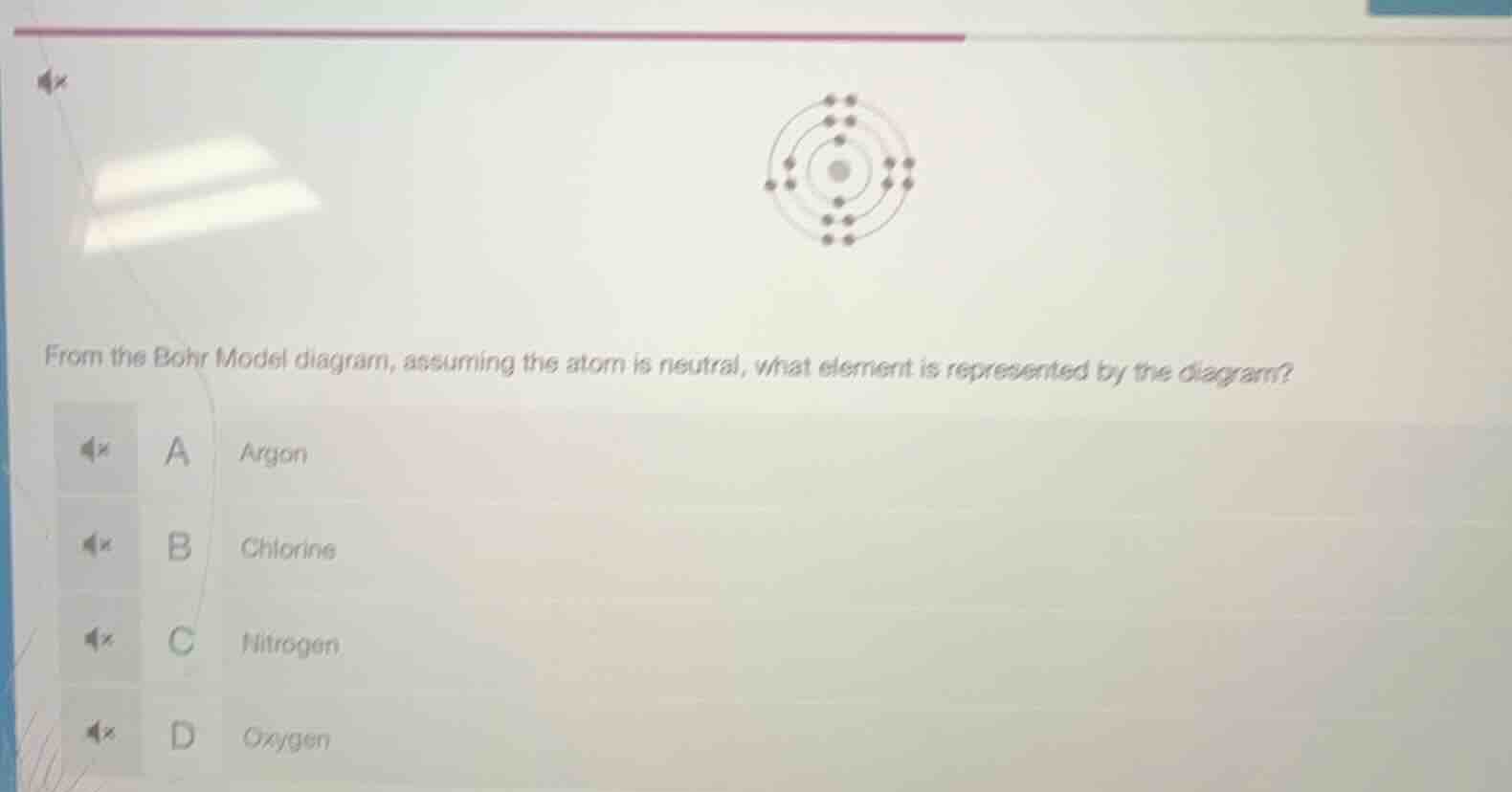
from the bohr model diagram, assuming the atom is neutral, what element is represented by the diagram? a argon b chlorine c nitrogen d oxygen
Brief Explanations
To determine the element from the Bohr Model (neutral atom), we count the number of electrons (equal to protons in a neutral atom, which is the atomic number). Let's analyze the electron shells:
- First shell (closest to nucleus) can hold up to 2 electrons, second up to 8, third up to 8, etc.
- From the diagram (assuming the electron counts per shell: first shell: 2, second: 8, third: 8? Wait, no—wait, let's re - evaluate. Wait, no, maybe the diagram has: first shell (innermost) 2, second 8, third 8? No, wait, no—wait, the options: Argon (Ar) has atomic number 18 (electrons: 2, 8, 8). Chlorine (Cl) has atomic number 17 (electrons: 2, 8, 7). Nitrogen (N) has atomic number 7 (electrons: 2, 5). Oxygen (O) has atomic number 8 (electrons: 2, 6). Wait, maybe the diagram's electron shells: let's check the Bohr model structure. If the atom has 2 (first shell), 8 (second), 8 (third)? No, that would be Argon. Wait, but maybe I misread. Wait, no—wait, the options: Argon is a noble gas with electron configuration 2, 8, 8. Let's confirm:
- Argon (Ar): atomic number 18, electrons: 2 (K), 8 (L), 8 (M).
- Chlorine (Cl): 17 electrons: 2, 8, 7.
- Nitrogen (N): 7 electrons: 2, 5.
- Oxygen (O): 8 electrons: 2, 6.
If the Bohr model shows 2, 8, 8 electrons (three shells), then it's Argon. So the correct option is A.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
A. Argon