QUESTION IMAGE
Question
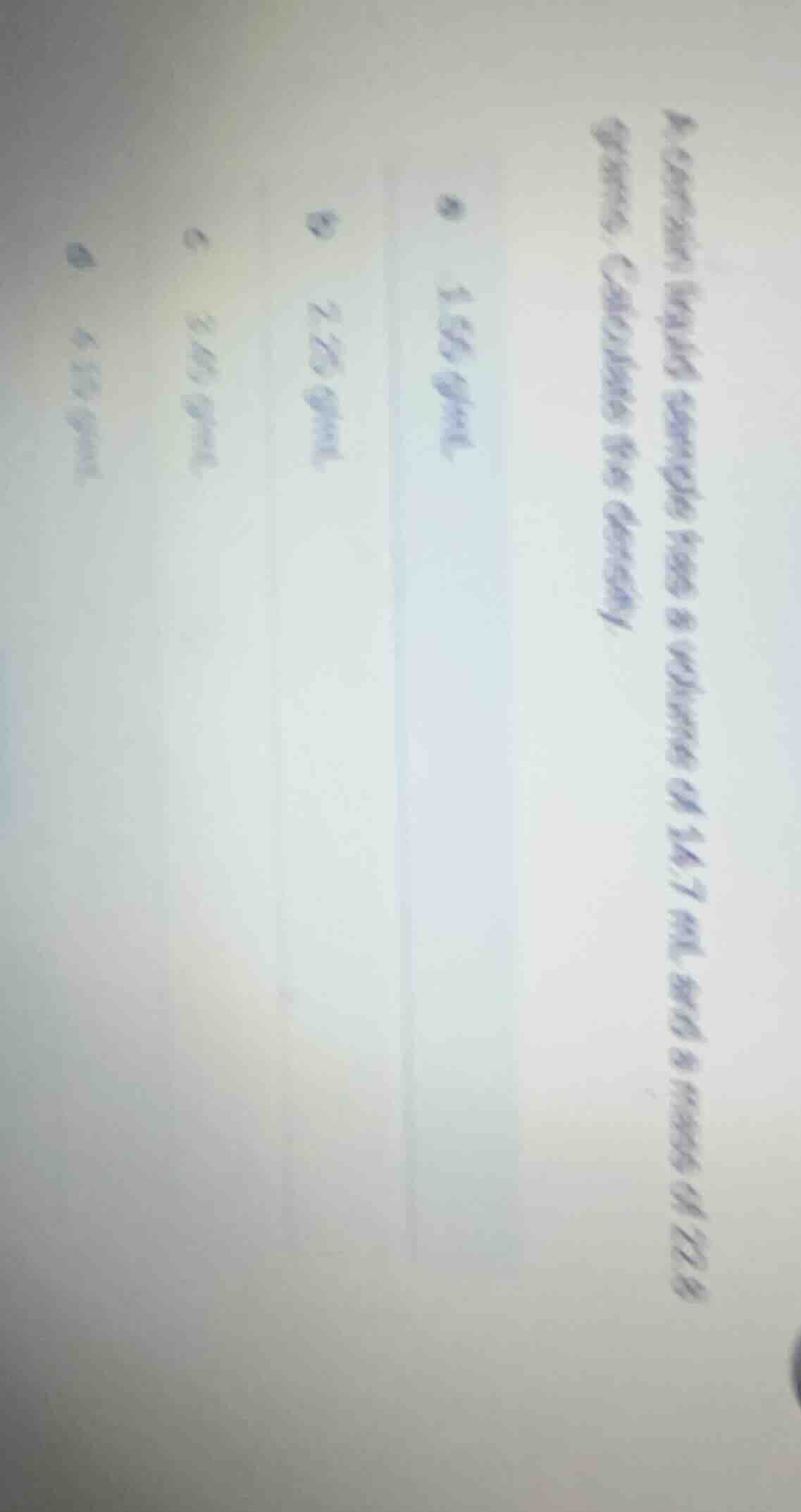
- a certain liquid sample has a volume of 14.7 ml and a mass of 22.8 grams. calculate its density. a 1.55 g/ml b 2.25 g/ml c 3.45 g/ml d 4.15 g/ml
Step1: Recall density formula
Density ($
ho$) is defined as mass ($m$) divided by volume ($V$), so the formula is $
ho = \frac{m}{V}$.
Step2: Identify given values
We are given the volume $V = 14.7\space mL$ and the mass $m = 22.8\space g$ (assuming the mass value is 22.8 g, as it's a common mass unit here).
Step3: Calculate density
Substitute the values into the formula: $
ho=\frac{22.8\space g}{14.7\space mL}$.
Calculating this division: $\frac{22.8}{14.7}\approx1.55\space g/mL$ (matching option a).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
a. 1.55 g/mL