QUESTION IMAGE
Question
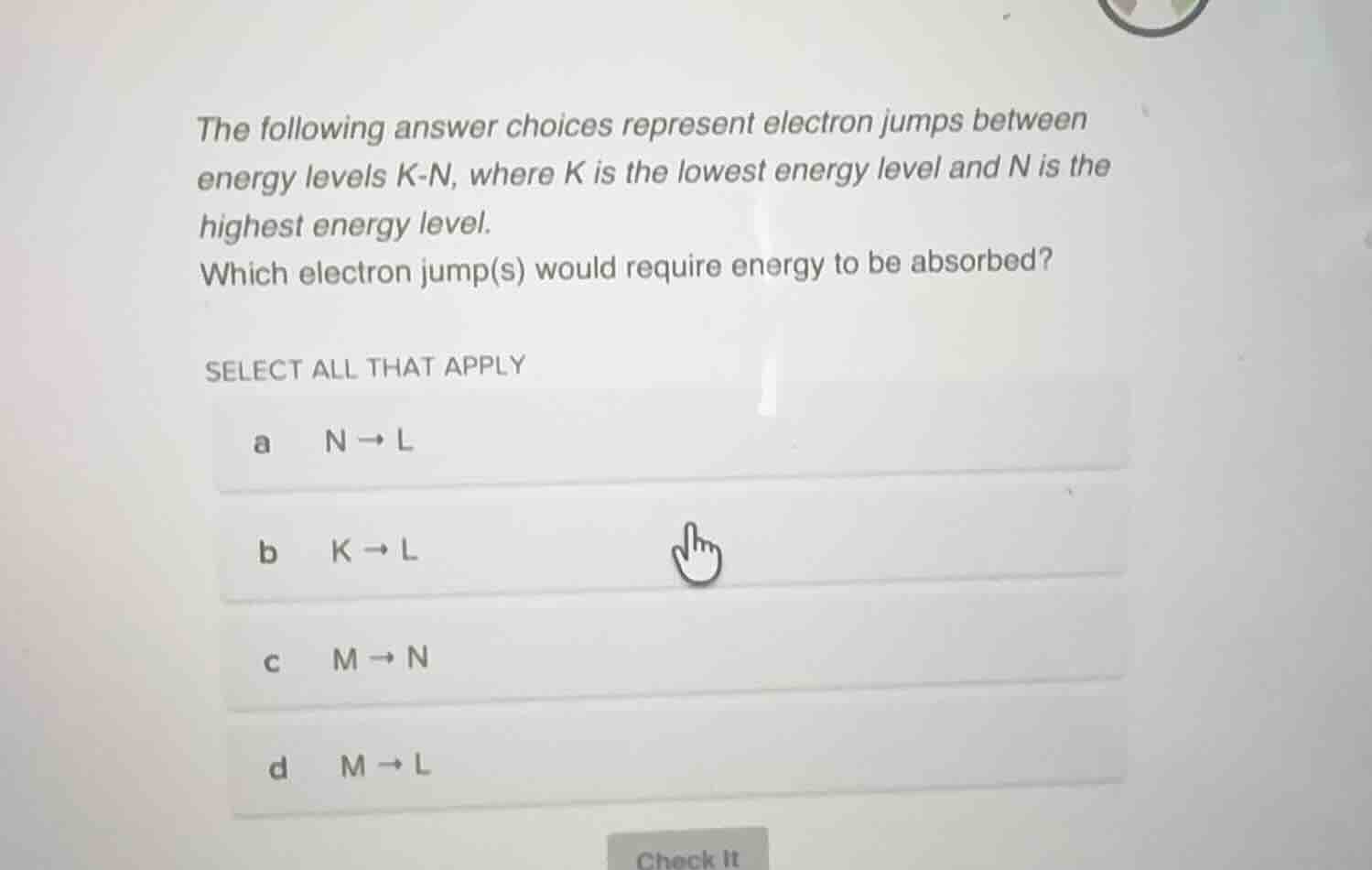
the following answer choices represent electron jumps between energy levels k-n, where k is the lowest energy level and n is the highest energy level. which electron jump(s) would require energy to be absorbed? select all that apply a n → l b k → l c m → n d m → l
To determine which electron jumps require energy absorption, we recall: Electrons absorb energy to move from a lower energy level to a higher energy level (excitation). They release energy when moving from a higher to a lower level (relaxation).
- Option a: \( N \to L \) is a jump from a higher (\( N \)) to a lower (\( L \)) energy level. This is relaxation, so energy is released, not absorbed.
- Option b: \( K \to L \) is a jump from the lowest energy level (\( K \)) to a higher level (\( L \)). This is excitation, so energy is absorbed.
- Option c: \( M \to N \) is a jump from a lower (\( M \)) to a higher (\( N \)) energy level. This is excitation, so energy is absorbed.
- Option d: \( M \to L \) is a jump from a higher (\( M \)) to a lower (\( L \)) energy level. This is relaxation, so energy is released, not absorbed.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
b. \( K \to L \), c. \( M \to N \)