QUESTION IMAGE
Question
the number of protons in an atom
○ atomic mass
○ atomic number
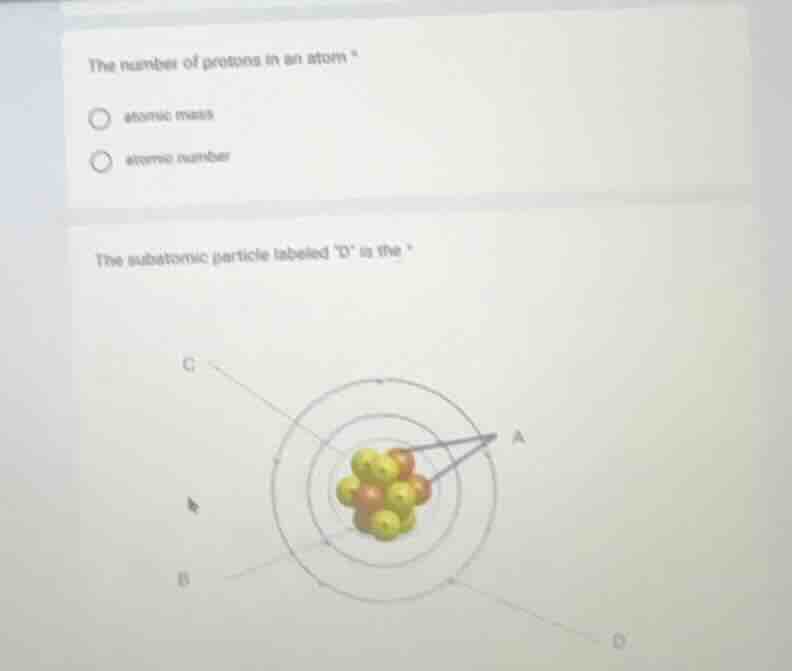
the subatomic particle labeled \o\ is the
First Sub - Question (The number of protons in an atom)
The atomic number of an atom is defined as the number of protons in its nucleus. Atomic mass is the sum of protons and neutrons in the nucleus. So the correct option related to the number of protons is atomic number.
In a typical atomic model diagram: A is usually protons/neutrons (nucleus), B is electrons (orbiting), D (assuming the diagram has D as an electron - like particle orbiting or a subatomic particle in the electron cloud region) - electrons are the subatomic particles that orbit the nucleus. If D is in the electron cloud (orbital) region, it's an electron. But since the diagram's labels: C is likely nucleus (protons/neutrons), A is protons/neutrons, B and D (if D is an electron) - electrons are negatively charged and orbit the nucleus. So the subatomic particle labeled D is an electron (assuming standard atomic model where electrons are in the electron cloud around the nucleus).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
atomic number (the option with "atomic number" text)