QUESTION IMAGE
Question
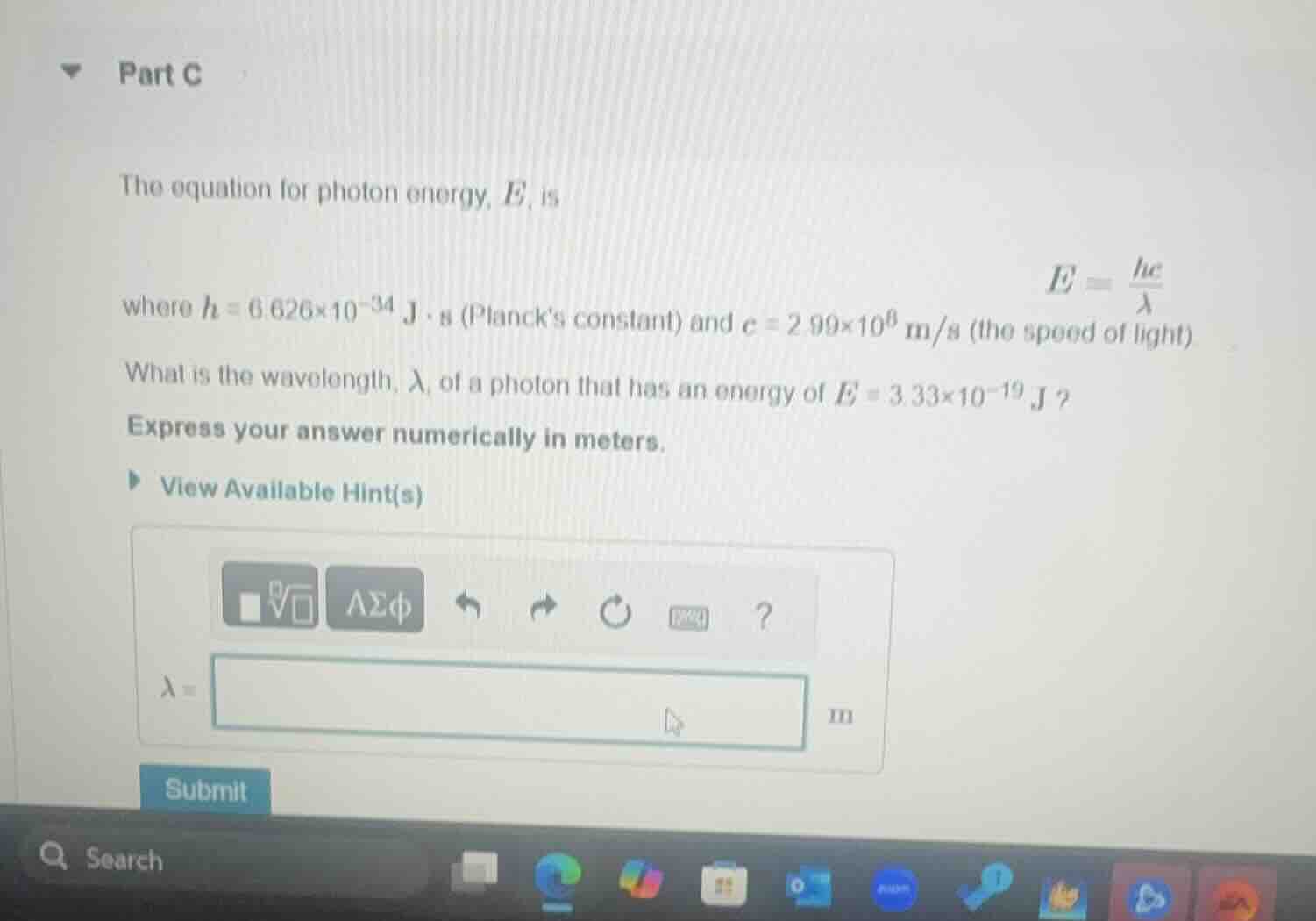
part c
the equation for photon energy, e, is
e = \frac{hc}{\lambda}
where h = 6.626×10⁻³⁴ j·s (planck’s constant) and c = 2.99×10⁸ m/s (the speed of light)
what is the wavelength, λ, of a photon that has an energy of e = 3.33×10⁻¹⁹ j?
express your answer numerically in meters.
view available hint(s)
λ = m
submit
Step1: Rearrange for $\lambda$
$\lambda = \frac{hc}{E}$
Step2: Substitute given values
$\lambda = \frac{(6.626 \times 10^{-34}\ \text{J·s})(2.99 \times 10^8\ \text{m/s})}{3.33 \times 10^{-19}\ \text{J}}$
Step3: Calculate numerator first
$(6.626 \times 10^{-34})(2.99 \times 10^8) = 1.981174 \times 10^{-25}$
Step4: Divide by energy value
$\lambda = \frac{1.981174 \times 10^{-25}}{3.33 \times 10^{-19}}$
Step5: Compute final value
$\lambda = 5.95 \times 10^{-7}$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
$5.95 \times 10^{-7}$ m