QUESTION IMAGE
Question
a particle with a positive or negative net charge is called an ion.
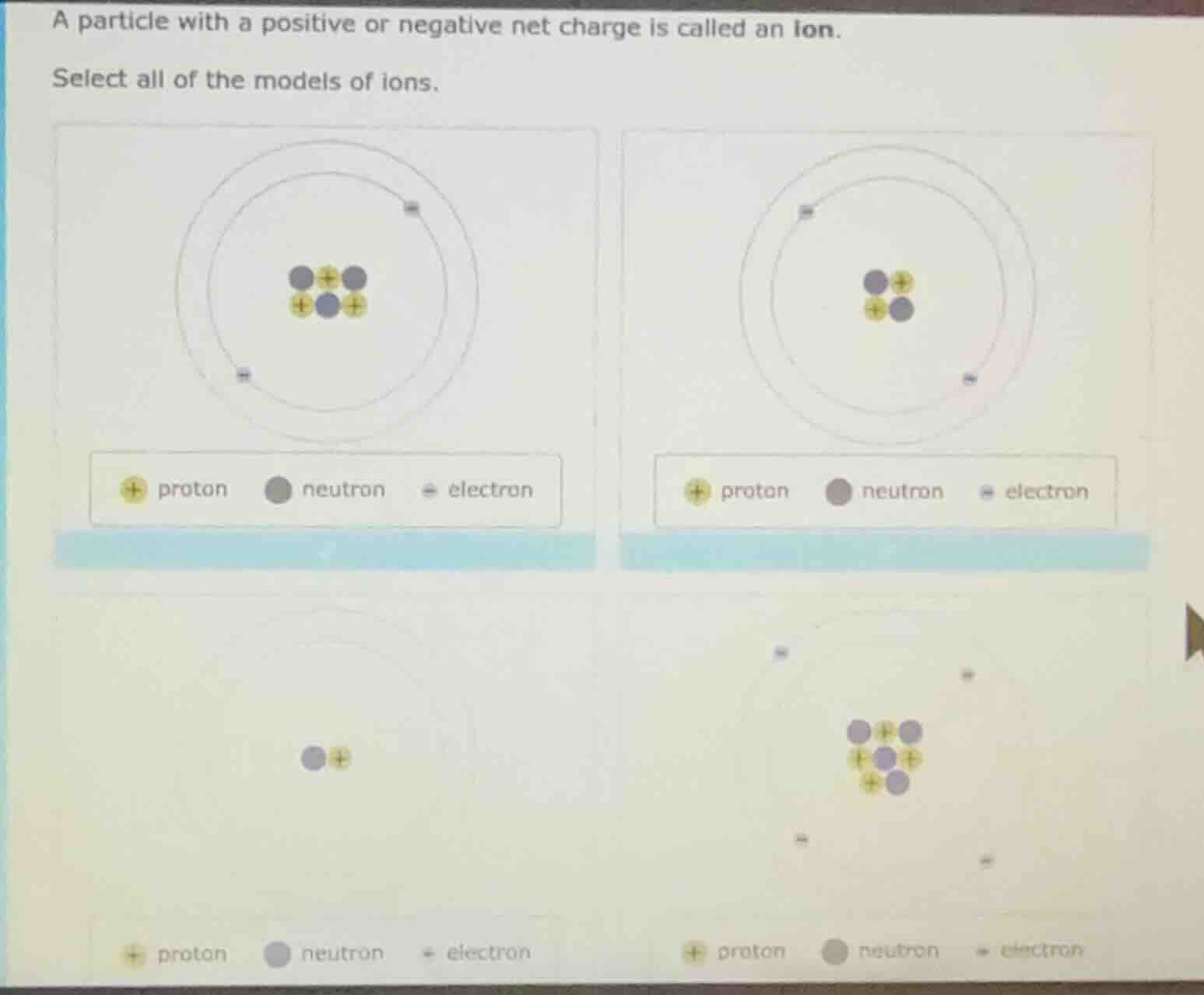
select all of the models of ions.
four atomic models with proton, neutron, electron legends are shown, but the specific details of the lower two models are partially unclear from the ocr; the upper two have proton (yellow +), neutron (gray circle), electron (gray -) legends, with the first upper model having 3 protons, 2 neutrons, 2 electrons; the second upper model having 2 protons, 2 neutrons, 2 electrons? (exact counts may vary based on image, but the key is identifying ion models by charge balance)
To determine if a model represents an ion, we check if the number of protons (positive charge) and electrons (negative charge) are unequal (ions have a net charge, so \( \text{protons}
eq \text{electrons} \)).
- Top - Left Model: Count protons (\( p \)) = 3, neutrons (\( n \)) = 2, electrons (\( e \)) = 2. \( p
eq e \) (3 ≠ 2), so it is an ion.
- Top - Right Model: \( p = 2 \), \( n = 2 \), \( e = 2 \). \( p = e \) (2 = 2), so it is a neutral atom (not an ion).
- Bottom - Left Model: \( p = 1 \), \( n = 1 \), \( e = 0 \). \( p
eq e \) (1 ≠ 0), so it is an ion.
- Bottom - Right Model: \( p = 4 \), \( n = 3 \), \( e = 4 \). \( p = e \) (4 = 4), so it is a neutral atom (not an ion).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
The models of ions are the top - left model, the bottom - left model. (If we assume the top - left is Model 1, top - right Model 2, bottom - left Model 3, bottom - right Model 4: Model 1, Model 3)