QUESTION IMAGE
Question
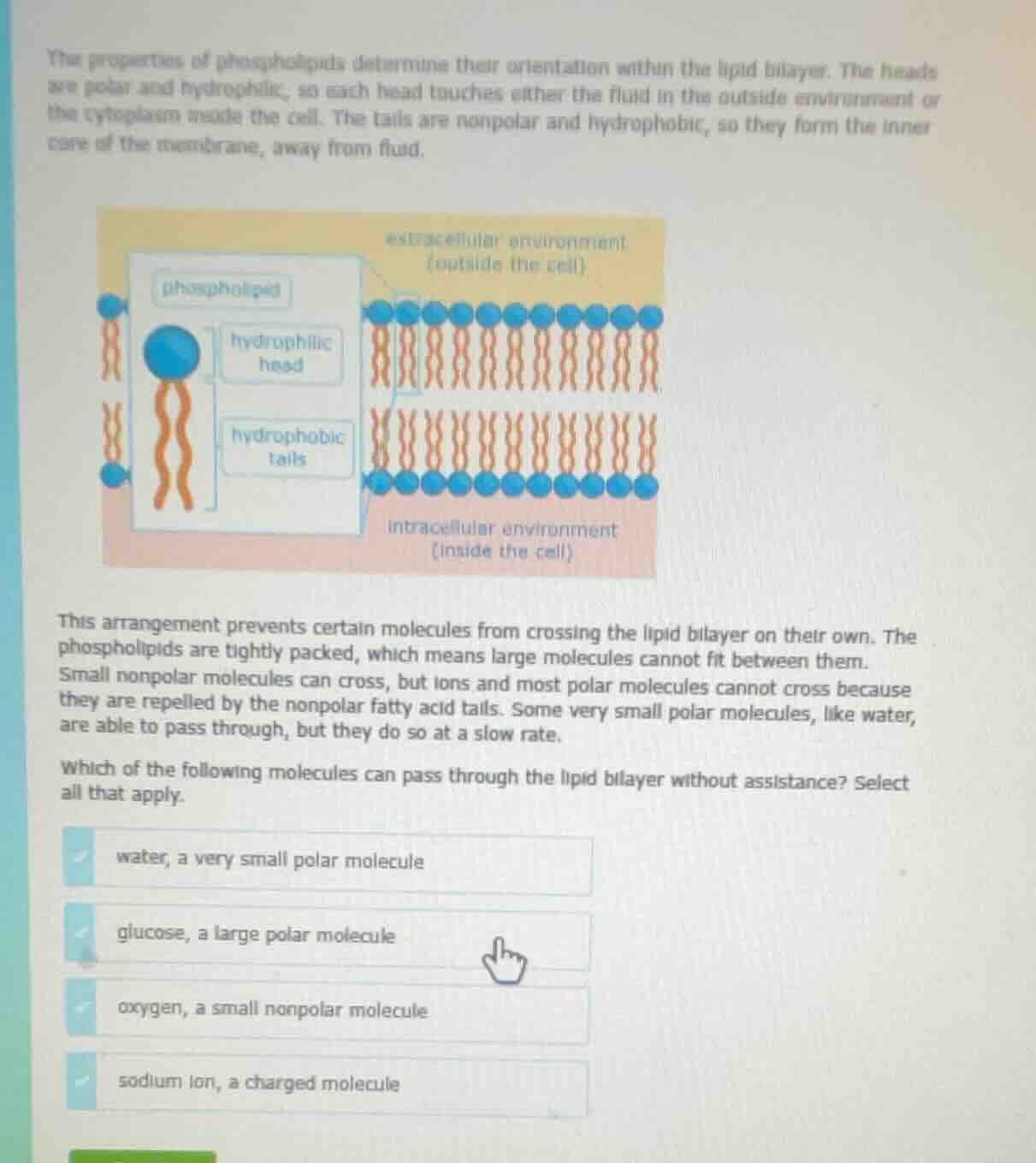
the properties of phospholipids determine their orientation within the lipid bilayer. the heads are polar and hydrophilic, so each head touches either the fluid in the outside environment or the cytoplasm inside the cell. the tails are nonpolar and hydrophobic, so they form the inner core of the membrane, away from fluid.
image of phospholipid bilayer structure with extracellular (outside the cell) and intracellular (inside the cell) environments, showing hydrophilic heads and hydrophobic tails
this arrangement prevents certain molecules from crossing the lipid bilayer on their own. the phospholipids are tightly packed, which means large molecules cannot fit between them. small nonpolar molecules can cross, but ions and most polar molecules cannot cross because they are repelled by the nonpolar fatty acid tails. some very small polar molecules, like water, are able to pass through, but they do so at a slow rate.
which of the following molecules can pass through the lipid bilayer without assistance? select all that apply.
- water, a very small polar molecule
- glucose, a large polar molecule
- oxygen, a small nonpolar molecule
- sodium ion, a charged molecule
- For water: The text says "Some very small polar molecules, like water, are able to pass through, but they do so at a slow rate", so water can pass without assistance.
- For glucose: The text states "large molecules cannot fit between" the phospholipids and "most polar molecules cannot cross", so glucose (large polar) cannot pass without assistance.
- For oxygen: The text says "Small nonpolar molecules can cross", and oxygen is a small nonpolar molecule, so it can pass without assistance.
- For sodium ion: The text says "ions... cannot cross because they are repelled by the nonpolar fatty acid tails", so sodium ion (charged/ion) cannot pass without assistance.
So the correct options are "water, a very small polar molecule" and "oxygen, a small nonpolar molecule".
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- water, a very small polar molecule
- oxygen, a small nonpolar molecule