QUESTION IMAGE
Question
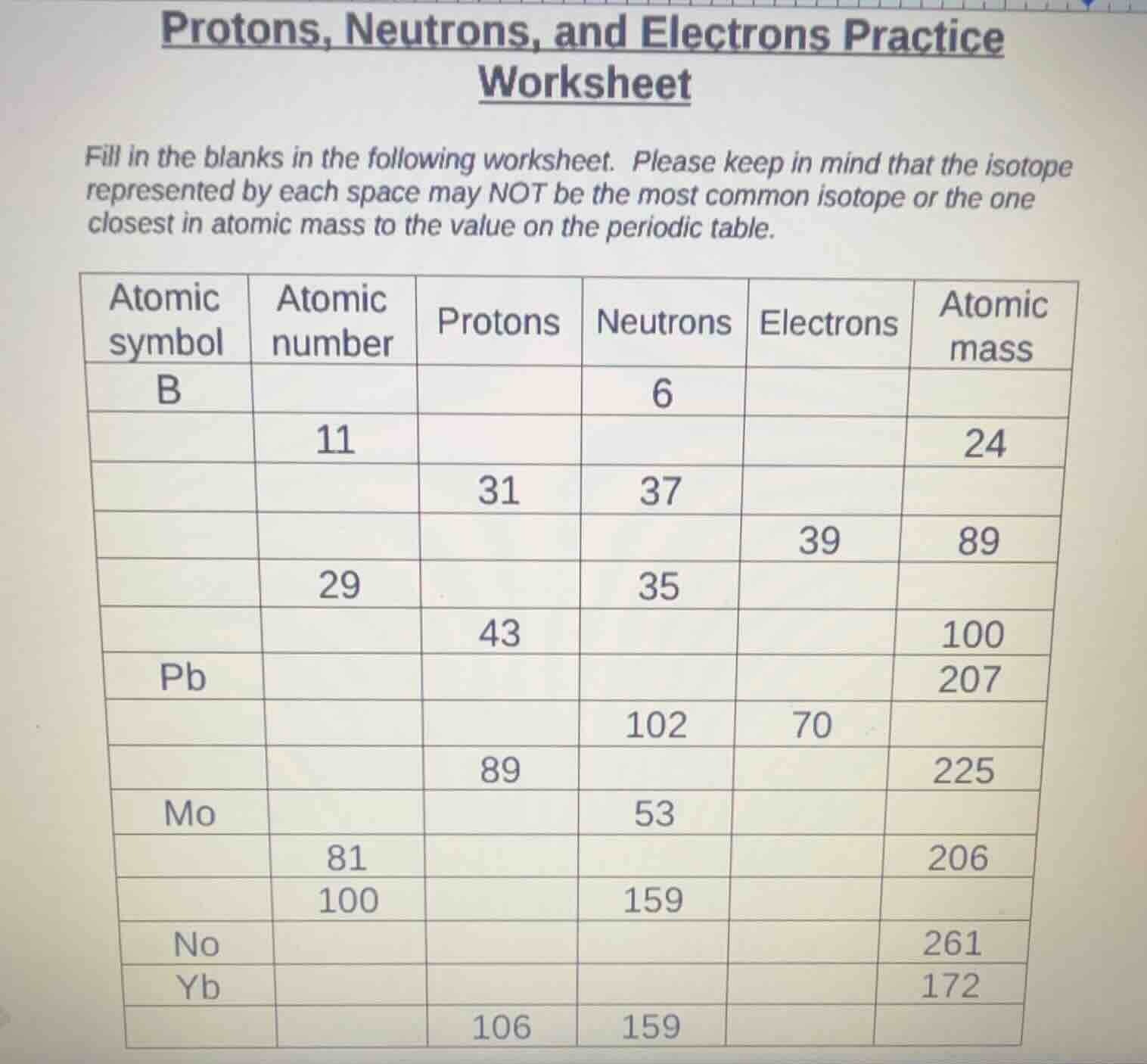
protons, neutrons, and electrons practice worksheet
fill in the blanks in the following worksheet. please keep in mind that the isotope represented by each space may not be the most common isotope or the one closest in atomic mass to the value on the periodic table.
| atomic symbol | atomic number | protons | neutrons | electrons | atomic mass |
|---|---|---|---|---|---|
| 11 | 24 | ||||
| 31 | 37 | ||||
| 39 | 89 | ||||
| 29 | 35 | ||||
| 43 | 100 | ||||
| pb | 207 | ||||
| 102 | 70 | ||||
| 89 | 225 | ||||
| mo | 53 | ||||
| 81 | 206 | ||||
| 100 | 159 | ||||
| no | 261 | ||||
| yb | 172 | ||||
| 106 | 159 |
Step1: Recall key atomic rules
- Atomic number = number of protons = number of electrons (neutral atom)
- Atomic mass = protons + neutrons
- Atomic symbol matches atomic number (periodic table)
---
Row 1 (Symbol B):
Step1: Find atomic number
Atomic number of B (boron) = 5, so protons = 5, electrons = 5
Step2: Calculate atomic mass
$\text{Atomic mass} = 5 + 6 = 11$
---
Row 2 (Atomic number 11, mass 24):
Step1: Identify symbol & protons
Atomic number 11 = Na (sodium); protons=11, electrons=11
Step2: Calculate neutrons
$\text{Neutrons} = 24 - 11 = 13$
---
Row 3 (Protons 31, neutrons 37):
Step1: Identify symbol & atomic number
Protons=31 = Ga (gallium); atomic number=31, electrons=31
Step2: Calculate atomic mass
$\text{Atomic mass} = 31 + 37 = 68$
---
Row 4 (Electrons 39, mass 89):
Step1: Find protons & atomic number
Electrons=39 = protons=39 = Y (yttrium); atomic number=39
Step2: Calculate neutrons
$\text{Neutrons} = 89 - 39 = 50$
---
Row 5 (Atomic number 29, neutrons 35):
Step1: Identify symbol & protons
Atomic number 29 = Cu (copper); protons=29, electrons=29
Step2: Calculate atomic mass
$\text{Atomic mass} = 29 + 35 = 64$
---
Row 6 (Protons 43, mass 100):
Step1: Identify symbol & atomic number
Protons=43 = Tc (technetium); atomic number=43, electrons=43
Step2: Calculate neutrons
$\text{Neutrons} = 100 - 43 = 57$
---
Row 7 (Symbol Pb, mass 207):
Step1: Find atomic number
Atomic number of Pb (lead) = 82; protons=82, electrons=82
Step2: Calculate neutrons
$\text{Neutrons} = 207 - 82 = 125$
---
Row 8 (Neutrons 102, electrons 70):
Step1: Find protons & atomic number
Electrons=70 = protons=70 = Yb (ytterbium); atomic number=70
Step2: Calculate atomic mass
$\text{Atomic mass} = 70 + 102 = 172$
---
Row 9 (Protons 89, mass 225):
Step1: Identify symbol & atomic number
Protons=89 = Ac (actinium); atomic number=89, electrons=89
Step2: Calculate neutrons
$\text{Neutrons} = 225 - 89 = 136$
---
Row 10 (Symbol Mo, neutrons 53):
Step1: Find atomic number
Atomic number of Mo (molybdenum) = 42; protons=42, electrons=42
Step2: Calculate atomic mass
$\text{Atomic mass} = 42 + 53 = 95$
---
Row 11 (Atomic number 81, mass 206):
Step1: Identify symbol & protons
Atomic number 81 = Tl (thallium); protons=81, electrons=81
Step2: Calculate neutrons
$\text{Neutrons} = 206 - 81 = 125$
---
Row 12 (Atomic number 100, neutrons 159):
Step1: Identify symbol & protons
Atomic number 100 = Fm (fermium); protons=100, electrons=100
Step2: Calculate atomic mass
$\text{Atomic mass} = 100 + 159 = 259$
---
Row 13 (Symbol No, mass 261):
Step1: Find atomic number
Atomic number of No (nobelium) = 102; protons=102, electrons=102
Step2: Calculate neutrons
$\text{Neutrons} = 261 - 102 = 159$
---
Row 14 (Symbol Yb, mass 172):
Step1: Find atomic number
Atomic number of Yb (ytterbium) = 70; protons=70, electrons=70
Step2: Calculate neutrons
$\text{Neutrons} = 172 - 70 = 102$
---
Row 15 (Protons 106, neutrons 159):
Step1: Identify symbol & atomic number
Protons=106 = Sg (seaborgium); atomic number=106, electrons=106
Step2: Calculate atomic mass
$\text{Atomic mass} = 106 + 159 = 265$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
| Atomic symbol | Atomic number | Protons | Neutrons | Electrons | Atomic mass |
|---|---|---|---|---|---|
| Na | 11 | 11 | 13 | 11 | 24 |
| Ga | 31 | 31 | 37 | 31 | 68 |
| Y | 39 | 39 | 50 | 39 | 89 |
| Cu | 29 | 29 | 35 | 29 | 64 |
| Tc | 43 | 43 | 57 | 43 | 100 |
| Pb | 82 | 82 | 125 | 82 | 207 |
| Yb | 70 | 70 | 102 | 70 | 172 |
| Ac | 89 | 89 | 136 | 89 | 225 |
| Mo | 42 | 42 | 53 | 42 | 95 |
| Tl | 81 | 81 | 125 | 81 | 206 |
| Fm | 100 | 100 | 159 | 100 | 259 |
| No | 102 | 102 | 159 | 102 | 261 |
| Yb | 70 | 70 | 102 | 70 | 172 |
| Sg | 106 | 106 | 159 | 106 | 265 |