QUESTION IMAGE
Question
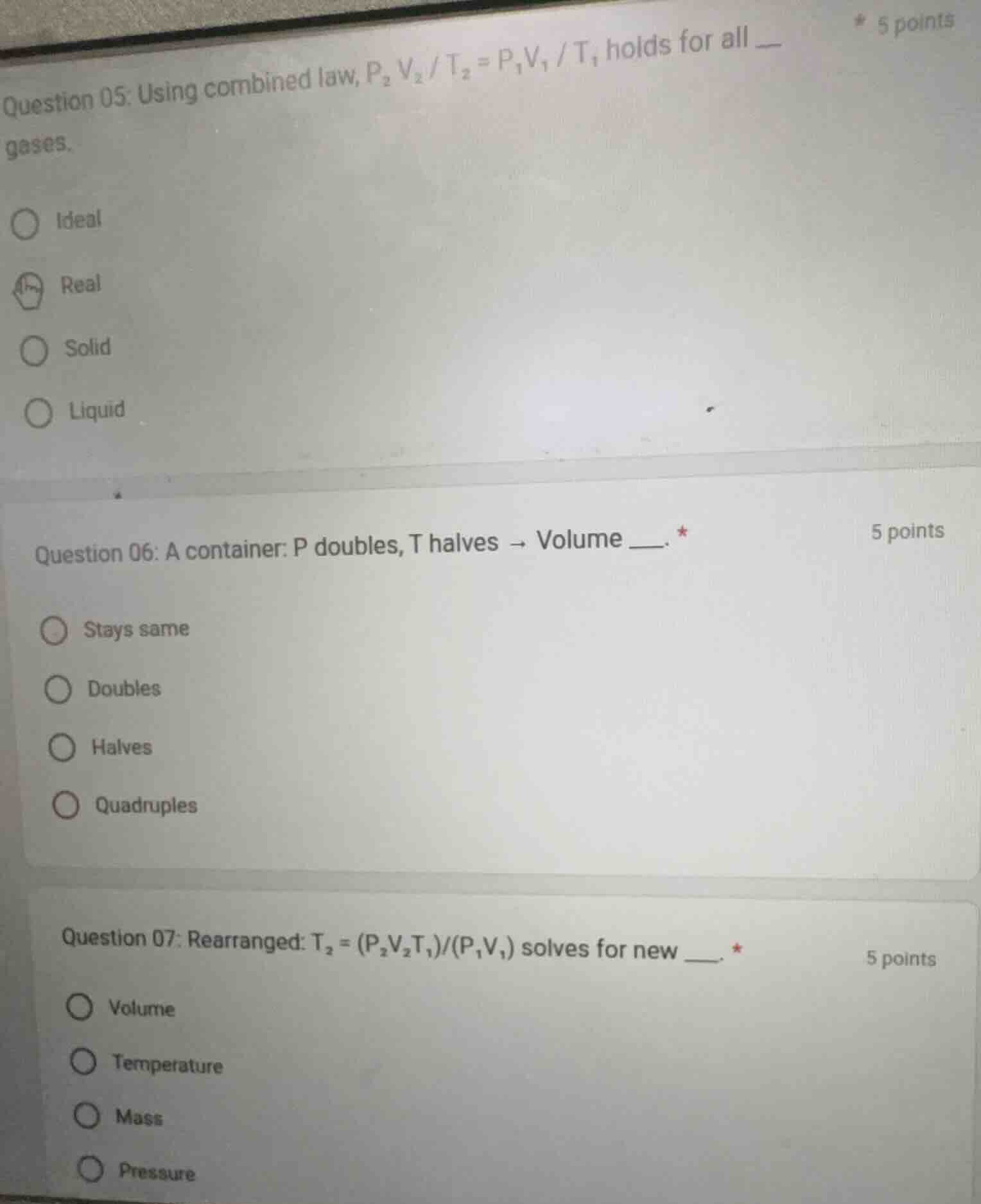
question 05: using combined law, $p_2v_2/t_2 = p_1v_1/t_1$ holds for all __ gases.
ideal
real
solid
liquid
question 06: a container: p doubles, t halves → volume __.
stays same
doubles
halves
quadruples
question 07: rearranged: $t_2 = (p_2v_2t_1)/(p_1v_1)$ solves for new __.
volume
temperature
mass
pressure
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Ideal
Quartered (Note: None of the provided options "Stays same", "Doubles", "Halves", "Quadruples" are correct; the volume becomes one-fourth of its original value.)
Temperature