QUESTION IMAGE
Question
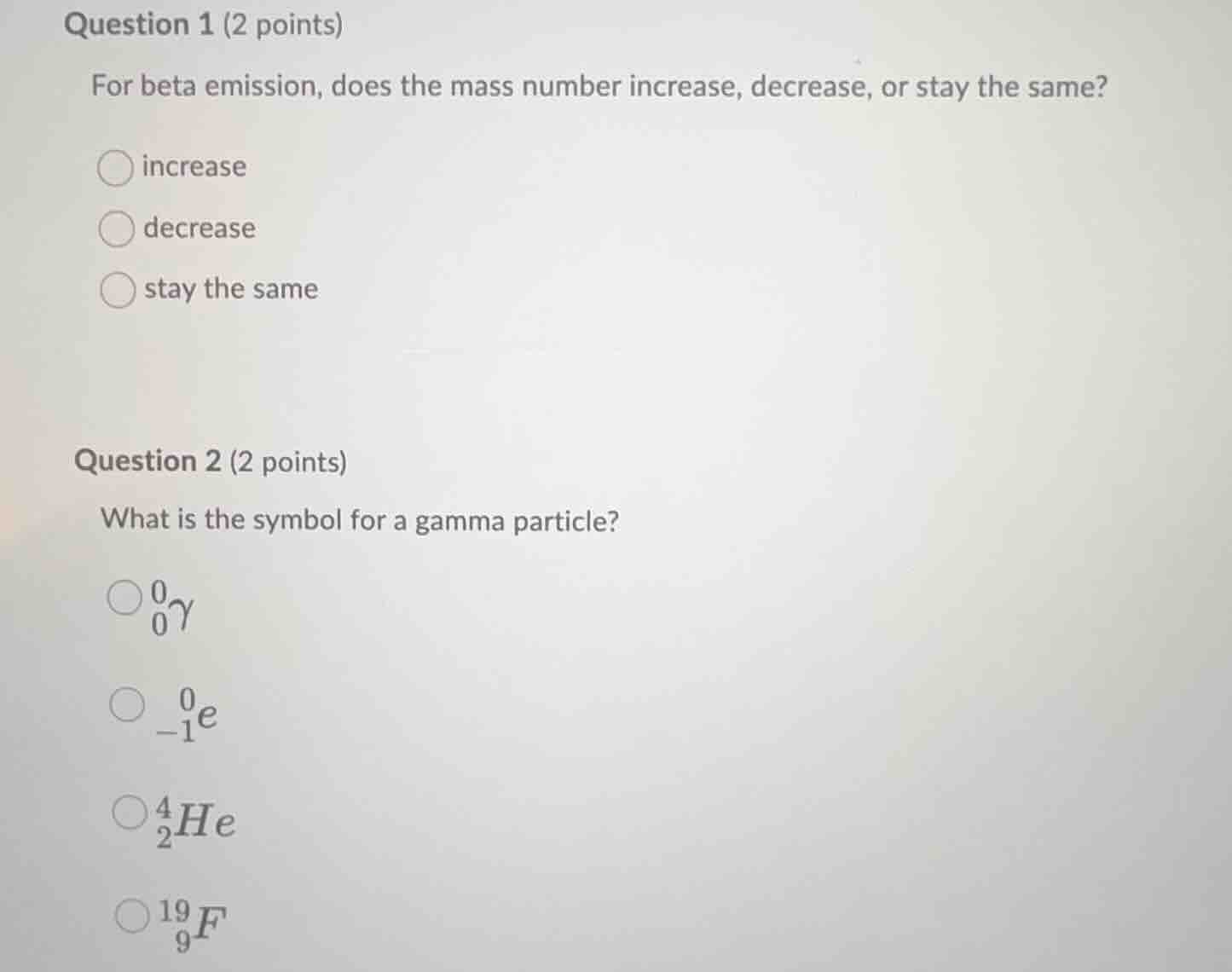
question 1 (2 points)
for beta emission, does the mass number increase, decrease, or stay the same?
○ increase
○ decrease
○ stay the same
question 2 (2 points)
what is the symbol for a gamma particle?
○ $\ce{_0^0\gamma}$
○ $\ce{_{-1}^0e}$
○ $\ce{_2^4he}$
○ $\ce{_9^{19}f}$
Question 1
In beta emission, a neutron in the nucleus converts to a proton and emits a beta particle ($\beta^-$ or $\beta^+$). The mass number is the sum of protons and neutrons. Since a neutron (mass number 1) is converted to a proton (mass number 1), the total number of nucleons (protons + neutrons) remains the same, so the mass number stays the same.
A gamma particle (photon) has no mass and no charge. Its symbol is $\ce{_0^0\gamma}$ because the mass number (top) and atomic number (bottom) are both 0 (no mass, no charge). $\ce{_{-1}^0e}$ is a beta particle, $\ce{_2^4He}$ is an alpha particle, and $\ce{_9^{19}F}$ is a fluorine atom.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
C. stay the same