QUESTION IMAGE
Question
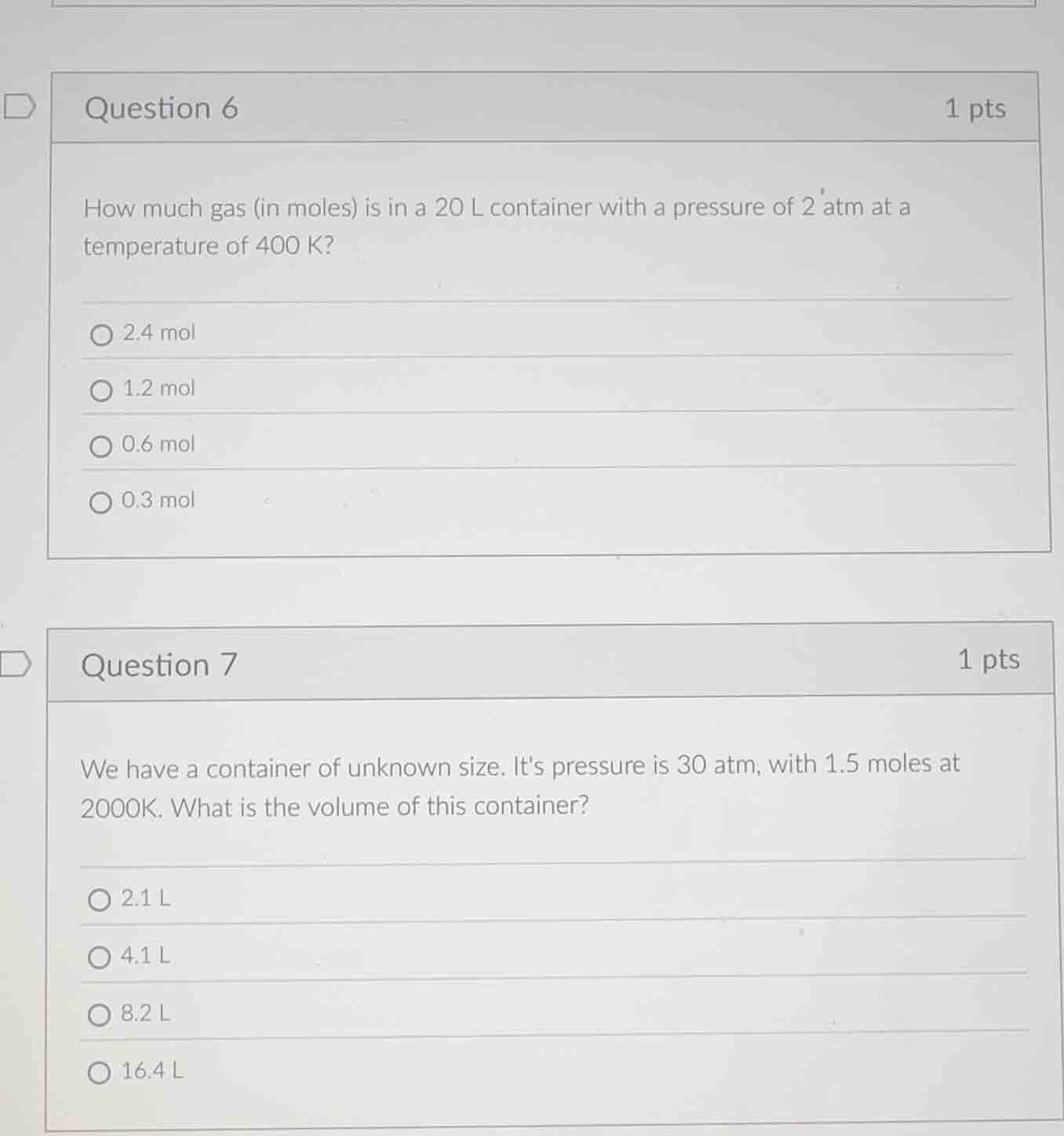
question 6 1 pts
how much gas (in moles) is in a 20 l container with a pressure of 2 atm at a temperature of 400 k?
2.4 mol
1.2 mol
0.6 mol
0.3 mol
question 7 1 pts
we have a container of unknown size. its pressure is 30 atm, with 1.5 moles at 2000k. what is the volume of this container?
2.1 l
4.1 l
8.2 l
16.4 l
Question 6
Step1: State ideal gas law rearranged
$n = \frac{PV}{RT}$
Step2: Substitute given values
$P=2\ \text{atm}, V=20\ \text{L}, R=0.0821\ \frac{\text{L·atm}}{\text{mol·K}}, T=400\ \text{K}$
$n = \frac{2 \times 20}{0.0821 \times 400}$
Step3: Calculate the result
$n = \frac{40}{32.84} \approx 1.2\ \text{mol}$
Question 7
Step1: State ideal gas law rearranged
$V = \frac{nRT}{P}$
Step2: Substitute given values
$n=1.5\ \text{mol}, R=0.0821\ \frac{\text{L·atm}}{\text{mol·K}}, T=2000\ \text{K}, P=30\ \text{atm}$
$V = \frac{1.5 \times 0.0821 \times 2000}{30}$
Step3: Calculate the result
$V = \frac{246.3}{30} = 8.21\ \text{L} \approx 8.2\ \text{L}$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Question 6: 1.2 mol
Question 7: 8.2 L