QUESTION IMAGE
Question
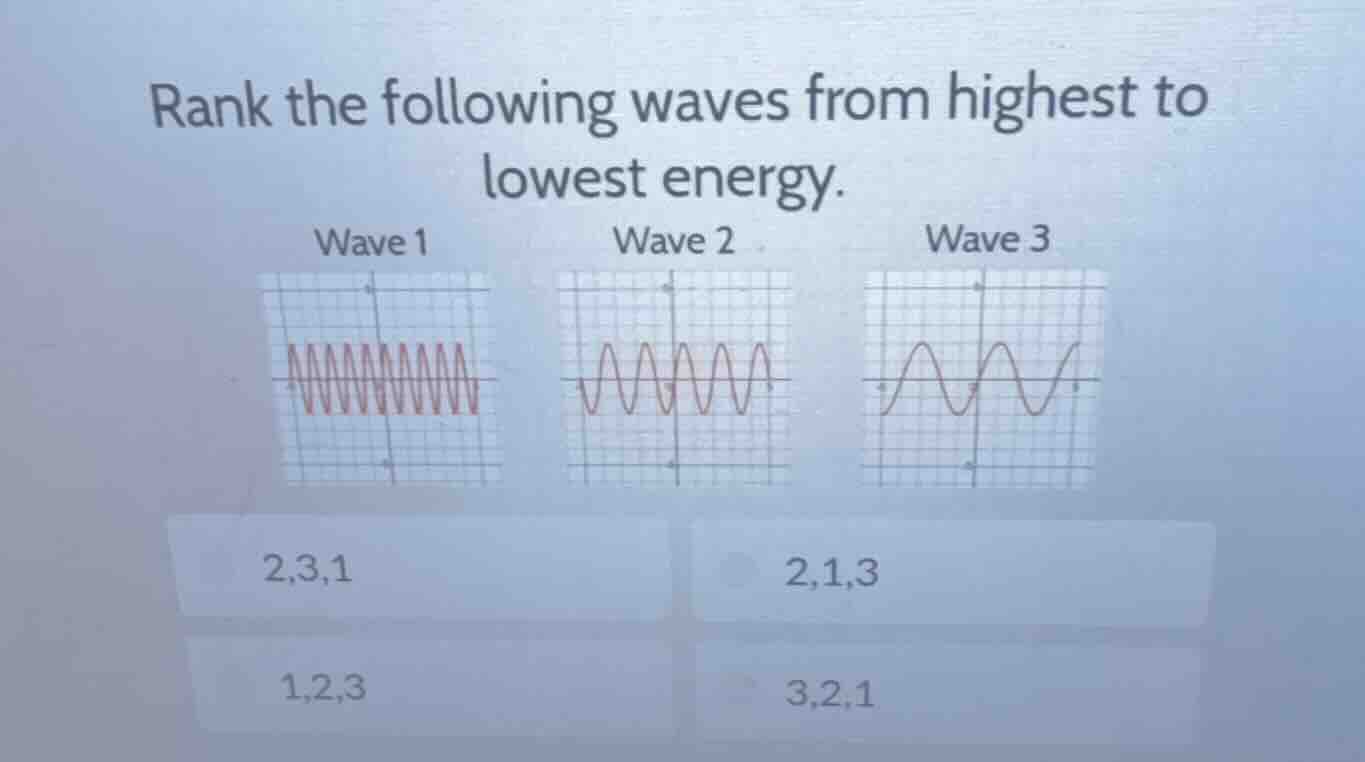
rank the following waves from highest to lowest energy. wave 1 wave 2 wave 3 2,3,1 2,1,3 1,2,3 3,2,1
Step1: Recall Energy - Frequency Relationship
The energy of a wave (for electromagnetic waves, or in general wave - particle duality context) is related to its frequency by the formula \(E = h
u\) (where \(h\) is Planck's constant and \(
u\) is the frequency). Higher frequency means higher energy. Frequency is the number of oscillations per unit time, which is related to the number of cycles (wavelengths) in a given distance. More cycles (higher frequency) mean higher energy.
Step2: Analyze the Waves' Frequencies
- Wave 1: Has the most number of cycles (oscillations) in the given grid space. So it has the highest frequency.
- Wave 2: Has fewer cycles than Wave 1 but more than Wave 3.
- Wave 3: Has the fewest number of cycles in the given grid space, so it has the lowest frequency.
Step3: Rank by Energy
Since energy is proportional to frequency (\(E\propto
u\)), the wave with the highest frequency has the highest energy and vice - versa. So the order from highest to lowest energy is Wave 1, Wave 2, Wave 3.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
1,2,3