QUESTION IMAGE
Question
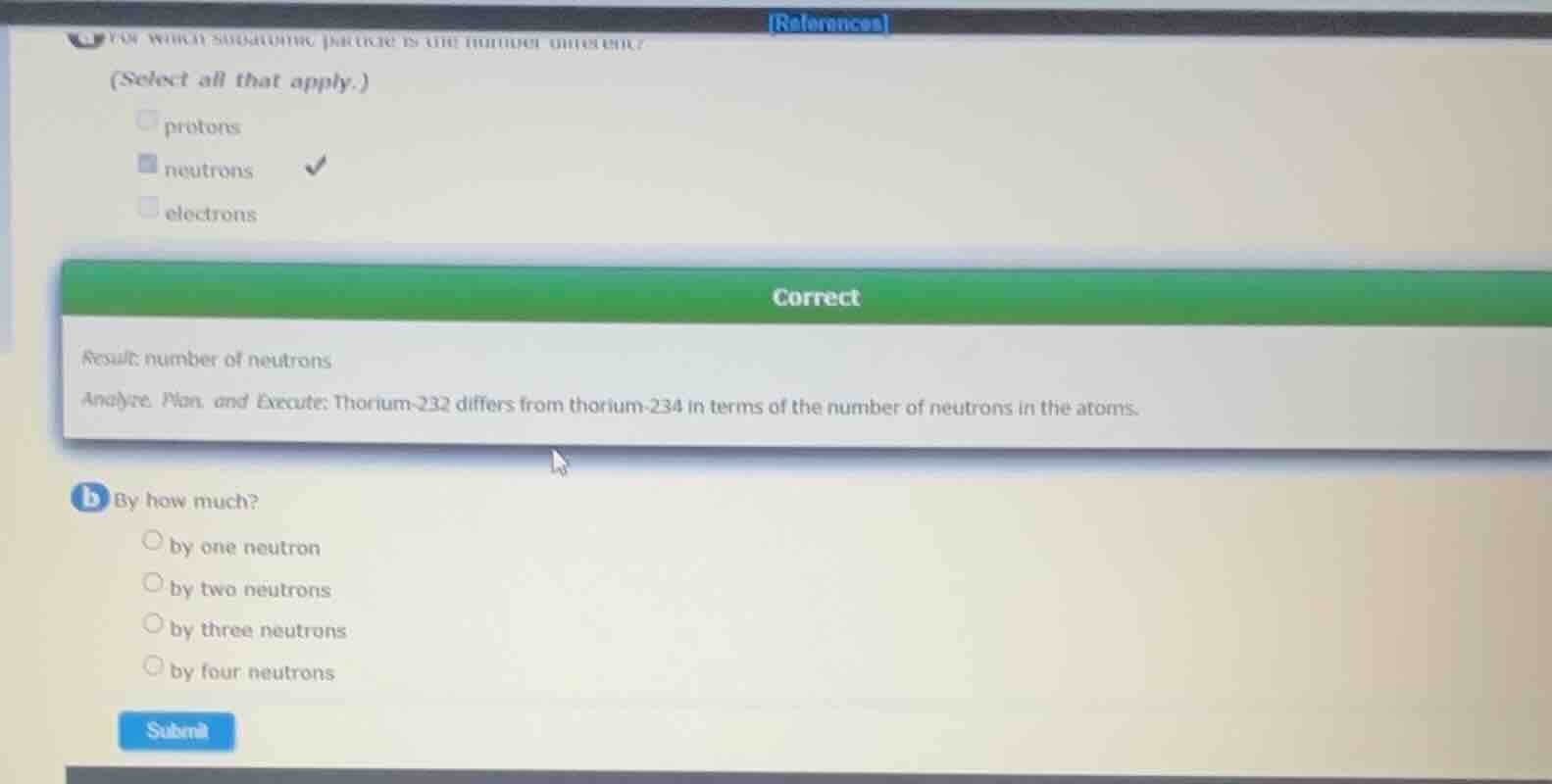
(select all that apply.) protons neutrons electrons correct result number of neutrons analyze, plan, and execute: thorium - 232 differs from thorium - 234 in terms of the number of neutrons in the atoms. b by how much? by one neutron by two neutrons by three neutrons by four neutrons submit
Step1: Recall the formula for neutrons
The number of neutrons in an atom is given by \( \text{Number of neutrons} = \text{Mass number} - \text{Atomic number} \). For thorium, the atomic number (number of protons) is constant (let's denote atomic number of Th as \( Z \)). Let the mass numbers be \( A_1 = 232 \) and \( A_2 = 234 \) for Th-232 and Th-234 respectively.
Step2: Calculate the difference in neutrons
The difference in the number of neutrons between Th-234 and Th-232 is \( (A_2 - Z) - (A_1 - Z) \). Simplifying this, the \( Z \) terms cancel out, so we get \( A_2 - A_1 \). Substituting the values, \( 234 - 232 = 2 \). So the difference in the number of neutrons is 2.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
by two neutrons