QUESTION IMAGE
Question
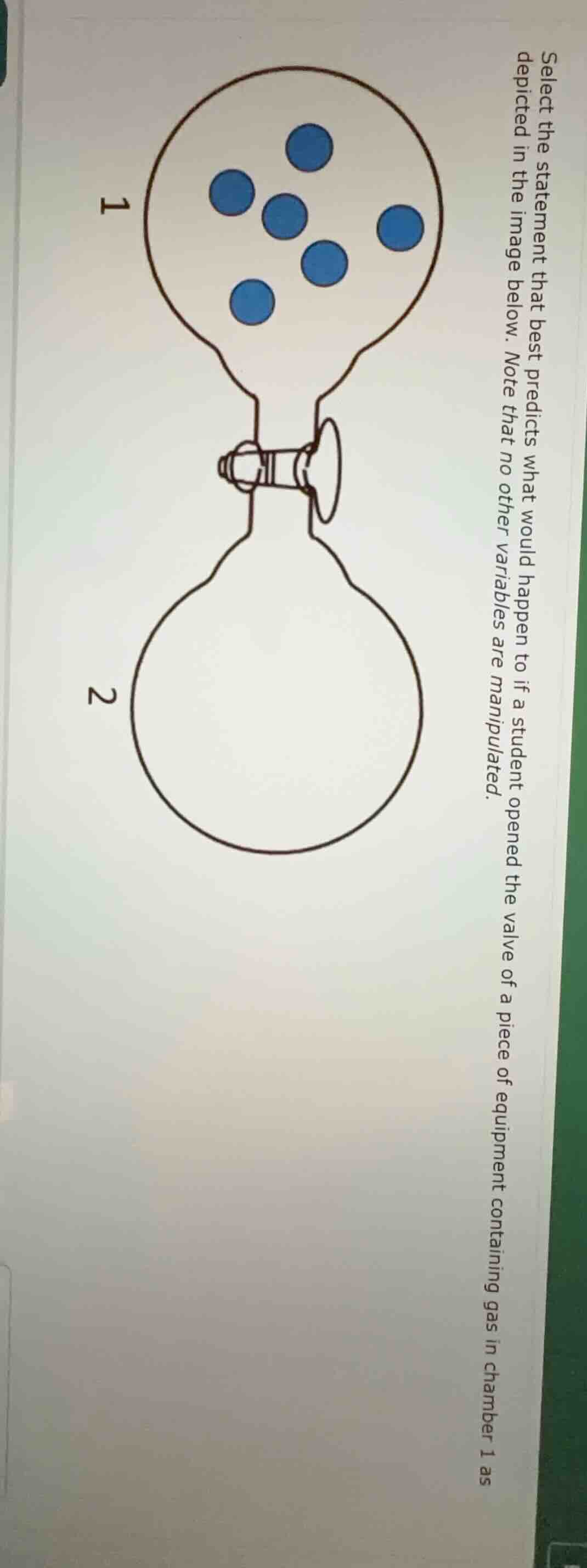
select the statement that best predicts what would happen if a student opened the valve of a piece of equipment containing gas in chamber 1 as depicted in the image below. note that no other variables are manipulated.
Since the problem is about gas behavior (likely related to gas laws, diffusion, or particle movement), which falls under Natural Science, specifically Physics (or Chemistry for gas properties). But as the question is to predict what happens when the valve is opened, we analyze:
Step1: Identify the system
Chamber 1 has gas particles (blue dots), Chamber 2 is empty. Valve connects them.
Step2: Apply particle theory/gas laws
Gas particles move randomly (kinetic theory). When valve opens, particles will diffuse from Chamber 1 (higher concentration) to Chamber 2 (lower concentration) until equilibrium (uniform distribution) is reached. So particles will spread into Chamber 2, and eventually, both chambers will have an even distribution of blue dots (assuming same volume/temperature, particles move to fill available space).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
When the valve is opened, the gas particles (blue dots) from Chamber 1 will diffuse into Chamber 2, and over time, the particles will be evenly distributed between both chambers as they move randomly to fill the available volume (due to the kinetic theory of gases and diffusion).