QUESTION IMAGE
Question
the structure of the atom
quiz complete
time remainin
59:20
1 2 3 4 5 6 7 8 9 10
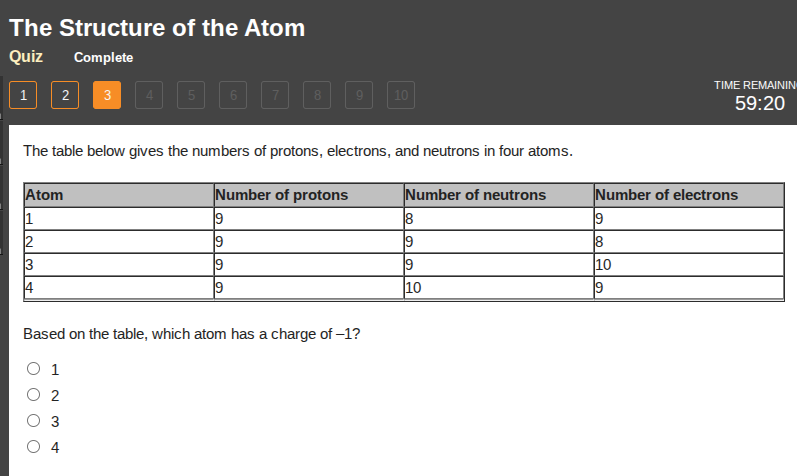
the table below gives the numbers of protons, electrons, and neutrons in four atoms.
atom | number of protons | number of neutrons | number of electrons
1 | 9 | 8 | 9
2 | 9 | 9 | 8
3 | 9 | 9 | 10
4 | 9 | 10 | 9
based on the table, which atom has a charge of -1?
○ 1
○ 2
○ 3
○ 4
Response
3: Calculate charge for Atom 2
$9 - 8 = +1$
Step4: Calculate charge for Atom 3
$9 - 10 = -1$
Step5: Calculate charge for Atom 4
$9 - 9 = 0$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
3: Calculate charge for Atom 2
$9 - 8 = +1$
Step4: Calculate charge for Atom 3
$9 - 10 = -1$
Step5: Calculate charge for Atom 4
$9 - 9 = 0$