QUESTION IMAGE
Question
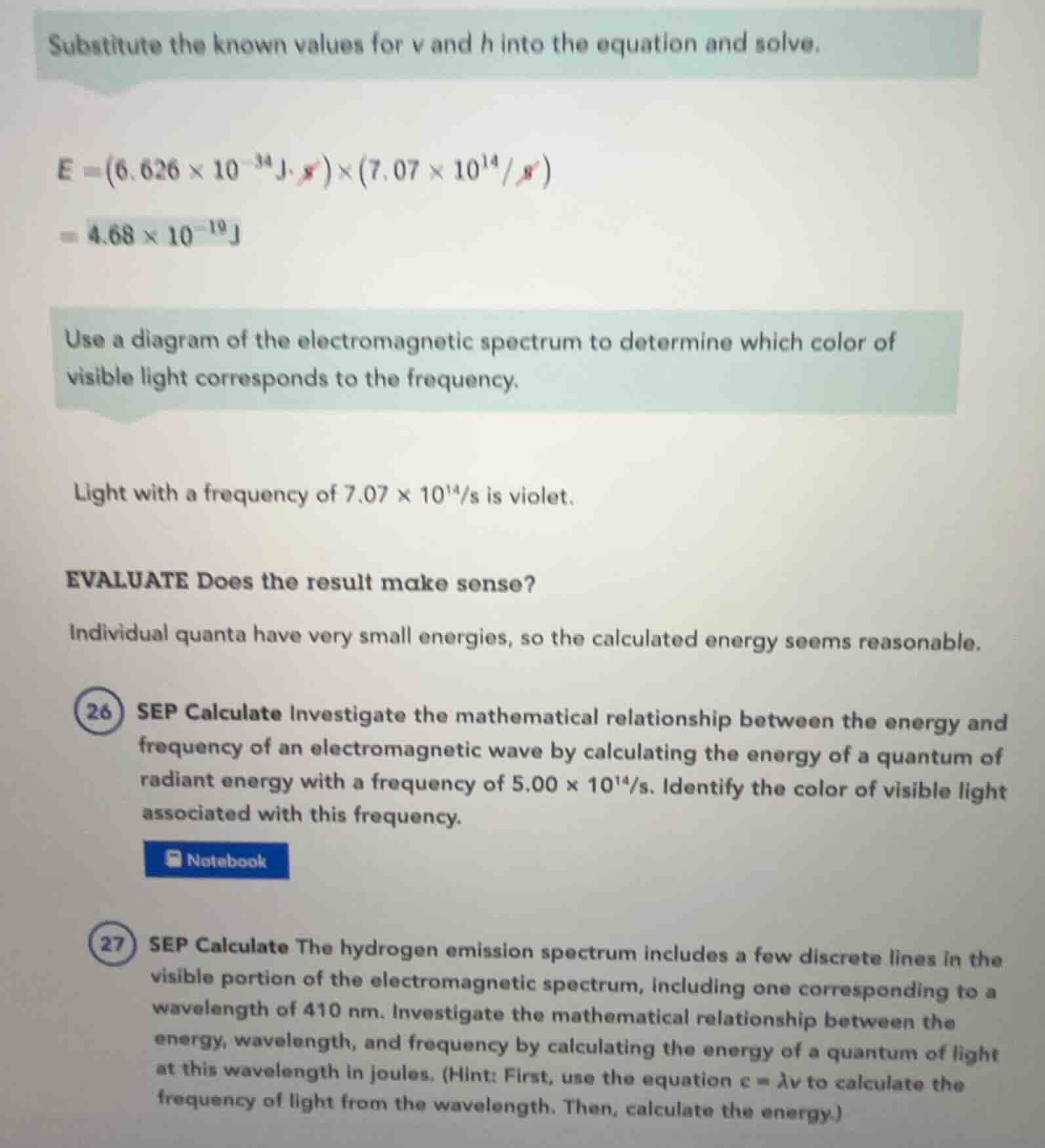
substitute the known values for v and h into the equation and solve.
$e = (6.626 \times 10^{-34} \text{j} \cdot \cancel{s}) \times (7.07 \times 10^{14} / \cancel{s})$
$= 4.68 \times 10^{-19} \text{j}$
use a diagram of the electromagnetic spectrum to determine which color of visible light corresponds to the frequency.
light with a frequency of $7.07 \times 10^{14}/s$ is violet.
evaluate does the result make sense?
individual quanta have very small energies, so the calculated energy seems reasonable.
26 sep calculate investigate the mathematical relationship between the energy and frequency of an electromagnetic wave by calculating the energy of a quantum of radiant energy with a frequency of $5.00 \times 10^{14}/s$. identify the color of visible light associated with this frequency.
notebook
27 sep calculate the hydrogen emission spectrum includes a few discrete lines in the visible portion of the electromagnetic spectrum, including one corresponding to a wavelength of 410 nm. investigate the mathematical relationship between the energy, wavelength, and frequency by calculating the energy of a quantum of light at this wavelength in joules. (hint: first, use the equation $c = \lambda v$ to calculate the frequency of light from the wavelength. then, calculate the energy.)
Problem 26
Step 1: Recall the energy - frequency formula
The formula for the energy \(E\) of a quantum of radiant energy is \(E = h
u\), where \(h = 6.626\times10^{- 34}\space J\cdot s\) (Planck's constant) and \(
u\) is the frequency.
Given \(
u=5.00\times 10^{14}\space /s\) and \(h = 6.626\times10^{-34}\space J\cdot s\)
Step 2: Substitute the values into the formula
\(E=(6.626\times 10^{-34}\space J\cdot s)\times(5.00\times 10^{14}\space /s)\)
First, multiply the coefficients: \(6.626\times5.00 = 33.13\)
Then, multiply the powers of 10: \(10^{-34}\times10^{14}=10^{-34 + 14}=10^{-20}\)
So, \(E = 33.13\times10^{-20}\space J=3.313\times 10^{-19}\space J\) (after re - writing in scientific notation)
Step 3: Determine the color of light
The visible light spectrum has the following approximate frequency ranges:
- Red: \(4.00 - 4.84\times10^{14}\space /s\)
- Orange: \(4.84 - 5.08\times10^{14}\space /s\)
- Yellow: \(5.08 - 5.26\times10^{14}\space /s\)
- Green: \(5.26 - 6.06\times10^{14}\space /s\)
- Blue: \(6.06 - 6.68\times10^{14}\space /s\)
- Violet: \(6.68 - 7.90\times10^{14}\space /s\)
The frequency \(
u = 5.00\times10^{14}\space /s\) lies in the range of green light (since \(5.00\times10^{14}\space /s\) is between \(5.08 - 5.26\times10^{14}\space /s\)? Wait, no, let's re - check the ranges. A more accurate range for green is \(5.26 - 6.06\times10^{14}\space Hz\) (where \(Hz = /s\)), and yellow is \(5.08 - 5.26\times10^{14}\space Hz\), and orange is \(4.84 - 5.08\times10^{14}\space Hz\). Our frequency is \(5.00\times10^{14}\space Hz\), which is in the orange - yellow border, but more precisely, the range for yellow is around \(5.1\times10^{14}-5.2\times10^{14}\space Hz\), and orange is \(4.8\times10^{14}-5.1\times10^{14}\space Hz\). So \(5.00\times10^{14}\space Hz\) is in the orange region? Wait, maybe my initial ranges were off. Let's use a more standard source: The frequency range for visible light is approximately \(4.0\times10^{14}\space Hz\) (red) to \(7.9\times10^{14}\space Hz\) (violet). The frequency of \(5.00\times10^{14}\space Hz\) is in the green - yellow? No, let's use the formula \(c=\lambda
u\) (where \(c = 3\times10^{8}\space m/s\)) to find the wavelength \(\lambda=\frac{c}{
u}\)
\(\lambda=\frac{3\times 10^{8}\space m/s}{5.00\times10^{14}\space /s}=6.0\times 10^{-7}\space m = 600\space nm\)
The wavelength range for green light is approximately \(495 - 570\space nm\), yellow is \(570 - 590\space nm\), orange is \(590 - 620\space nm\), red is \(620 - 750\space nm\), blue is \(450 - 495\space nm\), violet is \(400 - 450\space nm\)
\(\lambda = 600\space nm\) lies in the orange region (since \(590 - 620\space nm\) is orange). Wait, I think I made a mistake earlier. Let's recast:
If \(
u=5.00\times10^{14}\space /s\)
\(\lambda=\frac{c}{
u}=\frac{3\times10^{8}\space m/s}{5.00\times10^{14}\space /s}=6.0\times 10^{-7}\space m = 600\space nm\)
The wavelength of \(600\space nm\) is in the orange part of the visible spectrum (orange light has wavelengths around \(590 - 620\space nm\))
Problem 27
Step 1: Recall the formula \(c=\lambda
u\) to find frequency from wavelength
Given \(\lambda = 410\space nm=410\times10^{-9}\space m = 4.10\times10^{-7}\space m\) and \(c = 3.00\times10^{8}\space m/s\)
We can solve for \(
u\) from \(c=\lambda
u\), so \(
u=\frac{c}{\lambda}\)
Step 2: Calculate the frequency
\(
u=\frac{3.00\times10^{8}\space m/s}{4.10\times10^{-7}\space m}\)
First, divide the coefficients: \(\frac{3.00}{4.10}\approx0.7317\)
Then, divide the powers of 10: \(\frac{10^{8}}{10^{-7}}=10^{8+7}=10^{15}\)
So, \(
u\approx0.7317\times 10^{15}\space /s = 7.317\times10^{14}\space /s\)
Step 3: Use the energy - frequency formula \(E = h
u\) to find energy
Given \(h = 6.626\times10^{-34}\space J\cdot s\) and \(
u=7.317\times10^{14}\space /s\)
\(E=(6.626\times 10^{-34}\space J\cdot s)\times(7.317\times10^{14}\space /s)\)
Multiply the coefficients: \(6.626\times7.317\approx48.5\)
Multiply the powers of 10: \(10^{-34}\times10^{14}=10^{-20}\)
So, \(E\approx48.5\times10^{-20}\space J = 4.85\times10^{-19}\space J\) (after re - writing in scientific notation)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
(Problem 26):
The energy of the quantum is \(E = 3.31\times 10^{-19}\space J\) (rounded to three significant figures) and the color of light with frequency \(5.00\times10^{14}\space /s\) is orange (based on wavelength - frequency - color relationship).