QUESTION IMAGE
Question
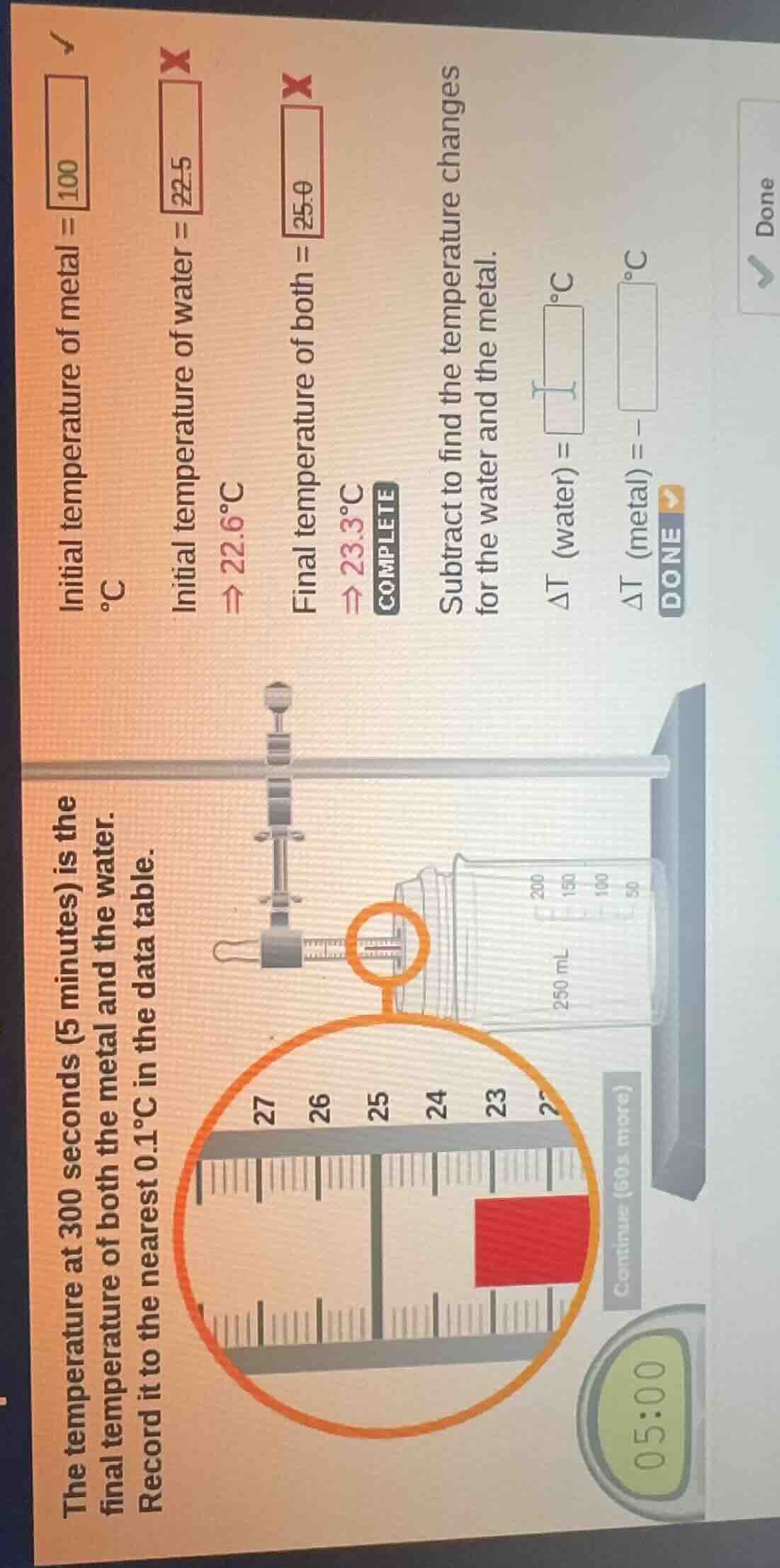
the temperature at 300 seconds (5 minutes) is the final temperature of both the metal and the water. record it to the nearest 0.1°c in the data table. initial temperature of metal = 100 °c initial temperature of water = 22.5 ⇒ 22.6°c final temperature of both = 25.0 ⇒ 23.3°c subtract to find the temperature changes for the water and the metal. δt (water) = \boxed{} °c δt (metal) = \boxed{} °c
Step1: Calculate ΔT for water
The formula for temperature change is $\Delta T = T_{final} - T_{initial}$. For water, $T_{initial} = 22.6^\circ\text{C}$ and $T_{final} = 23.3^\circ\text{C}$. So we calculate $23.3 - 22.6$.
$23.3 - 22.6 = 0.7$
Step2: Calculate ΔT for metal
For the metal, $T_{initial} = 100^\circ\text{C}$ and $T_{final} = 23.3^\circ\text{C}$. Using the same formula $\Delta T = T_{final} - T_{initial}$, we calculate $23.3 - 100$.
$23.3 - 100 = -76.7$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
$\Delta T$ (water) = $0.7^\circ\text{C}$
$\Delta T$ (metal) = $-76.7^\circ\text{C}$