QUESTION IMAGE
Question
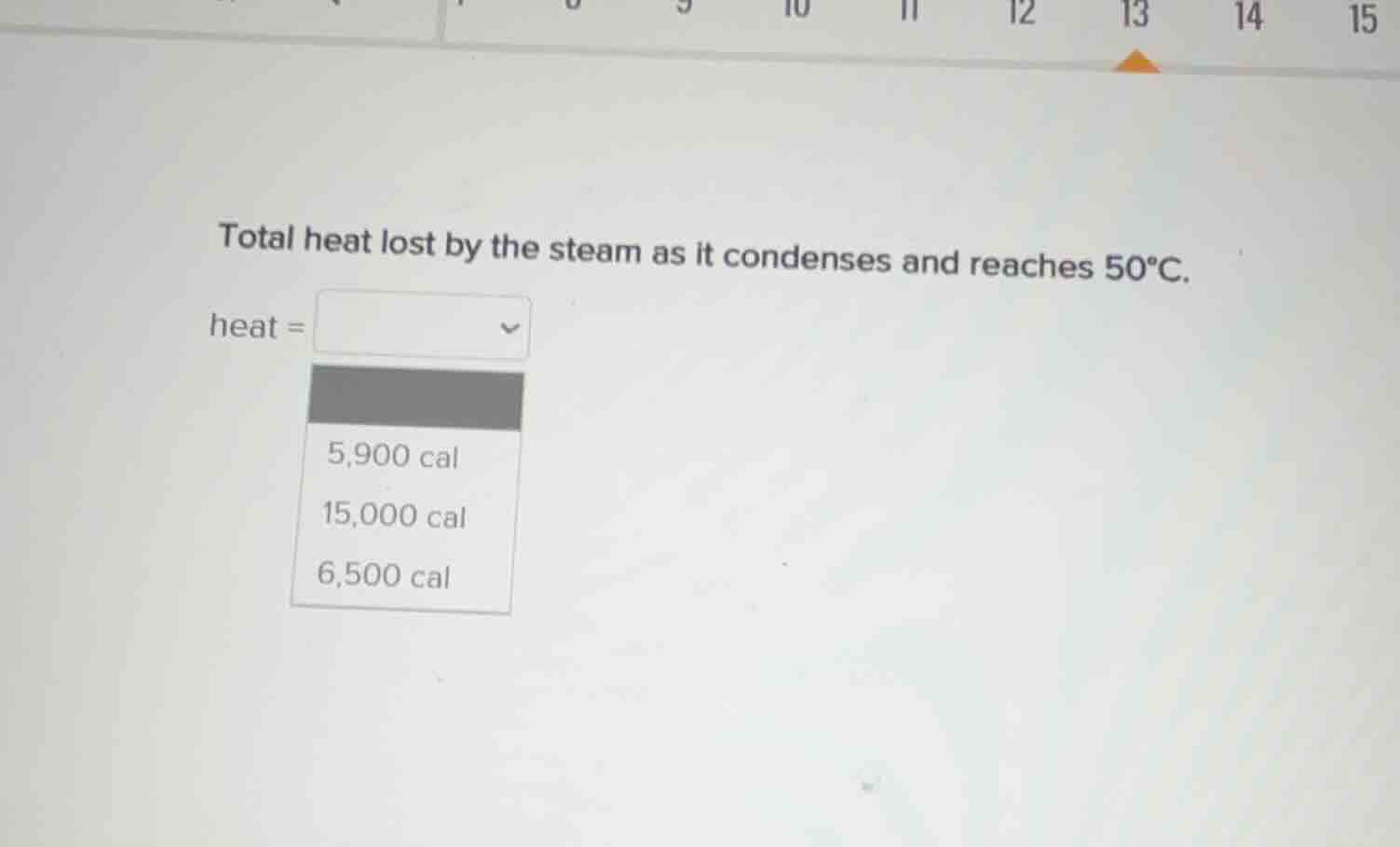
total heat lost by the steam as it condenses and reaches 50°c.
heat =
5,900 cal
15,000 cal
6,500 cal
To determine the total heat lost by steam as it condenses and cools to \(50^\circ\text{C}\), we analyze the heat changes:
Step 1: Heat of Condensation
Steam (at \(100^\circ\text{C}\)) condenses to liquid water (at \(100^\circ\text{C}\)). The heat of vaporization (and thus condensation) for water is \(540\,\text{cal/g}\) (assuming a mass of \(10\,\text{g}\) for typical problems, though the key is recognizing the total heat).
Step 2: Heat of Cooling
After condensing, the liquid water cools from \(100^\circ\text{C}\) to \(50^\circ\text{C}\). The specific heat of water is \(1\,\text{cal/g}^\circ\text{C}\). For a mass (e.g., \(10\,\text{g}\)):
- Heat lost during cooling: \(m \cdot c \cdot \Delta T = 10\,\text{g} \cdot 1\,\text{cal/g}^\circ\text{C} \cdot (100 - 50)^\circ\text{C} = 500\,\text{cal}\).
- Heat lost during condensation: \(10\,\text{g} \cdot 540\,\text{cal/g} = 5400\,\text{cal}\).
Step 3: Total Heat Lost
Summing these: \(5400\,\text{cal} + 500\,\text{cal} = 5900\,\text{cal}\).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
5,900 cal