QUESTION IMAGE
Question
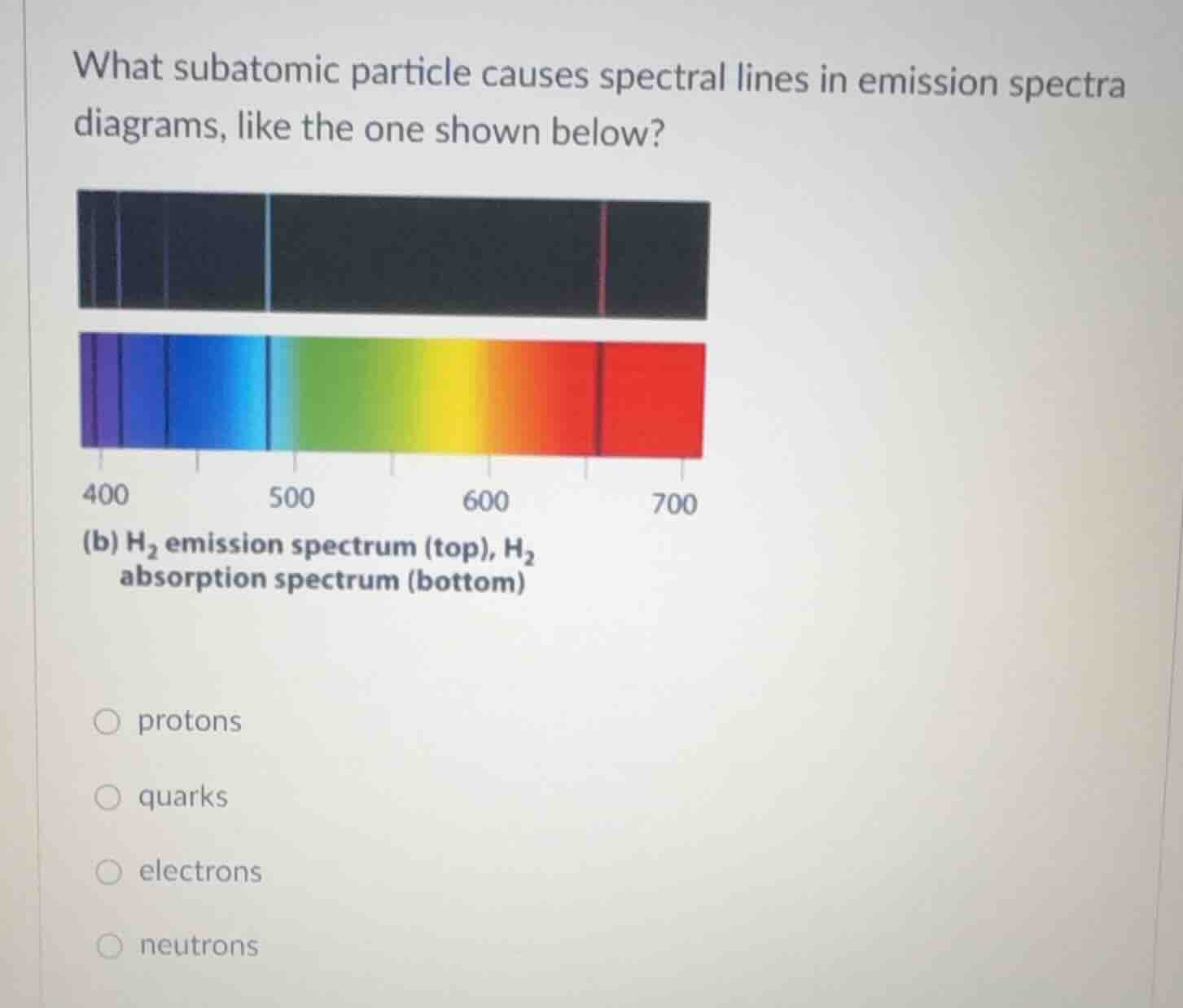
what subatomic particle causes spectral lines in emission spectra diagrams, like the one shown below?
(b) h₂ emission spectrum (top), h₂ absorption spectrum (bottom)
○ protons
○ quarks
○ electrons
○ neutrons
Spectral lines in emission spectra are caused by electrons transitioning between energy levels in atoms. When electrons move from higher to lower energy levels, they emit photons of specific energies (wavelengths), creating spectral lines. Protons, neutrons, and quarks do not directly cause spectral lines in this context.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
electrons (the option corresponding to "electrons")