QUESTION IMAGE
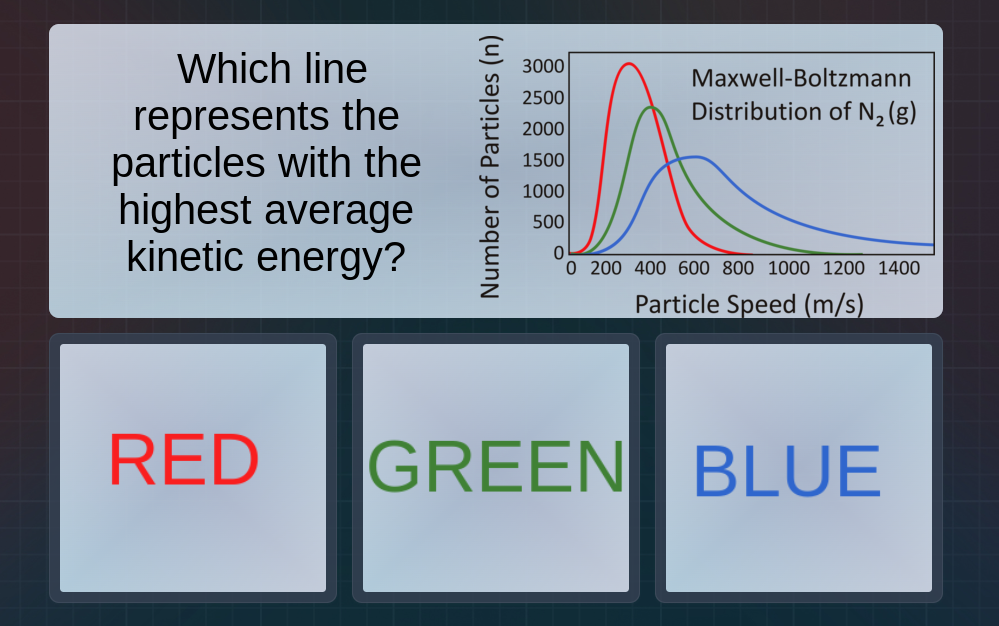
Question
which line represents the particles with the highest average kinetic energy? maxwell - boltzmann distribution of n₂(g) red green blue
The average kinetic energy of gas particles is related to temperature, and for a given gas (here \( \text{N}_2 \) gas), the distribution of particle speeds (Maxwell - Boltzmann distribution) shows that as the average speed of particles increases, the average kinetic energy (since \( KE=\frac{1}{2}mv^{2} \), for the same mass \( m \) of \( \text{N}_2 \) molecules, higher speed \( v \) means higher \( KE \)) increases. Looking at the Maxwell - Boltzmann distribution graph, the blue line is shifted towards higher particle speeds compared to red and green. So the line with the highest average kinetic energy is the blue one.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
BLUE