QUESTION IMAGE
Question
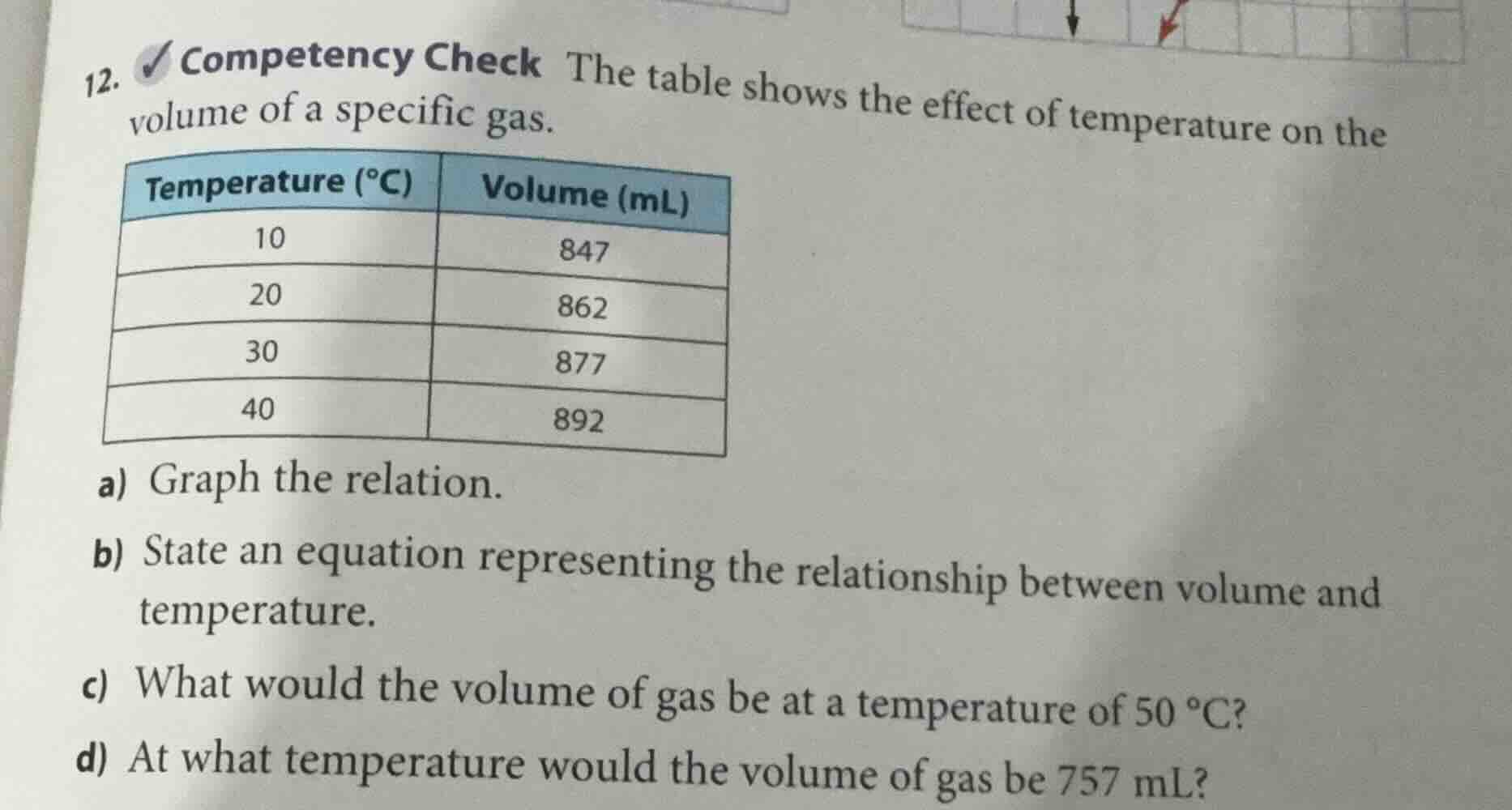
- competency check the table shows the effect of temperature on the volume of a specific gas.
| temperature (°c) | volume (ml) |
|---|---|
| 20 | 862 |
| 30 | 877 |
| 40 | 892 |
a) graph the relation.
b) state an equation representing the relationship between volume and temperature.
c) what would the volume of gas be at a temperature of 50 °c?
d) at what temperature would the volume of gas be 757 ml?
Step1: Identify linear relationship
First, confirm the relationship is linear by checking the constant rate of change. For temperature increase of 10°C, volume increases by 15 mL, so slope $m = \frac{15}{10} = 1.5$.
Step2: Find the y-intercept
Use point-slope form $V - V_1 = m(T - T_1)$ with $(T_1, V_1) = (10, 847)$:
$V - 847 = 1.5(T - 10)$
Simplify to slope-intercept form:
$V = 1.5T - 15 + 847$
$V = 1.5T + 832$
Step3: Calculate volume at 50°C
Substitute $T = 50$ into the equation:
$V = 1.5(50) + 832$
$V = 75 + 832$
Step4: Calculate temperature for 757 mL
Substitute $V = 757$ into the equation and solve for $T$:
$757 = 1.5T + 832$
$1.5T = 757 - 832$
$1.5T = -75$
$T = \frac{-75}{1.5}$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
a) (Graph: Plot the points (10, 847), (20, 862), (30, 877), (40, 892) on a coordinate plane with Temperature (°C) on the x-axis and Volume (mL) on the y-axis, then draw a straight line through them.)
b) $V = 1.5T + 832$
c) 907 mL
d) $-50^\circ\text{C}$