QUESTION IMAGE
Question
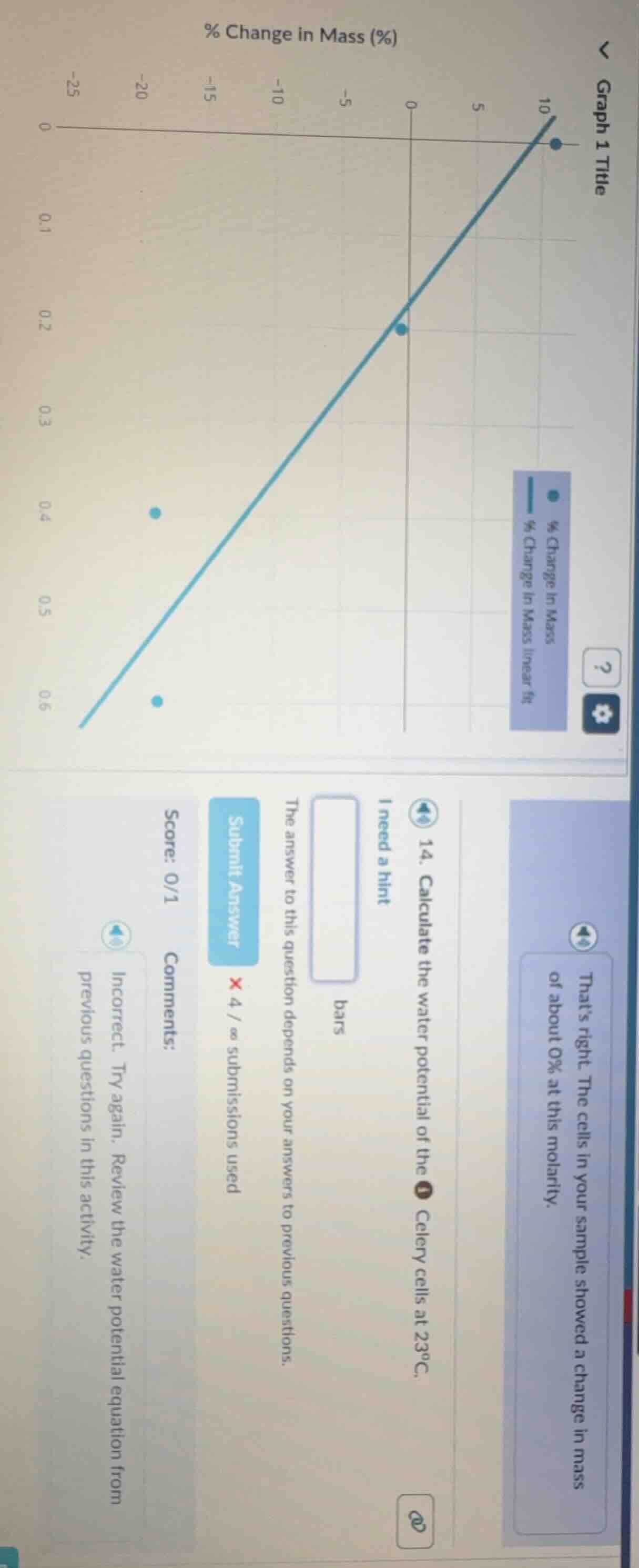
- calculate the water potential of the celery cells at 23°c. i need a hint the answer to this question depends on your answers to previous questions. submit answer × 4 / ∞ submissions used score: 0/1 comments: incorrect. try again. review the water potential equation from previous questions in this activity. thats right the cells in your sample showed a change in mass of about 0% at this molarity.
Step1: Find isotonic molarity
From the graph, the linear fit crosses 0% mass change at a molarity of $0.2\ \text{M}$.
Step2: Apply water potential formula
Use $\Psi = -iCRT$, where $i=1$ (sucrose, non-ionizing), $C=0.2\ \text{mol/L}$, $R=0.0831\ \text{bar·L/(mol·K)}$, $T=23+273=296\ \text{K}$.
$$\Psi = -1 \times 0.2 \times 0.0831 \times 296$$
Step3: Calculate the value
$$\Psi = -0.2 \times 0.0831 \times 296 = -4.92$$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
$-4.92$ bars