QUESTION IMAGE
Question
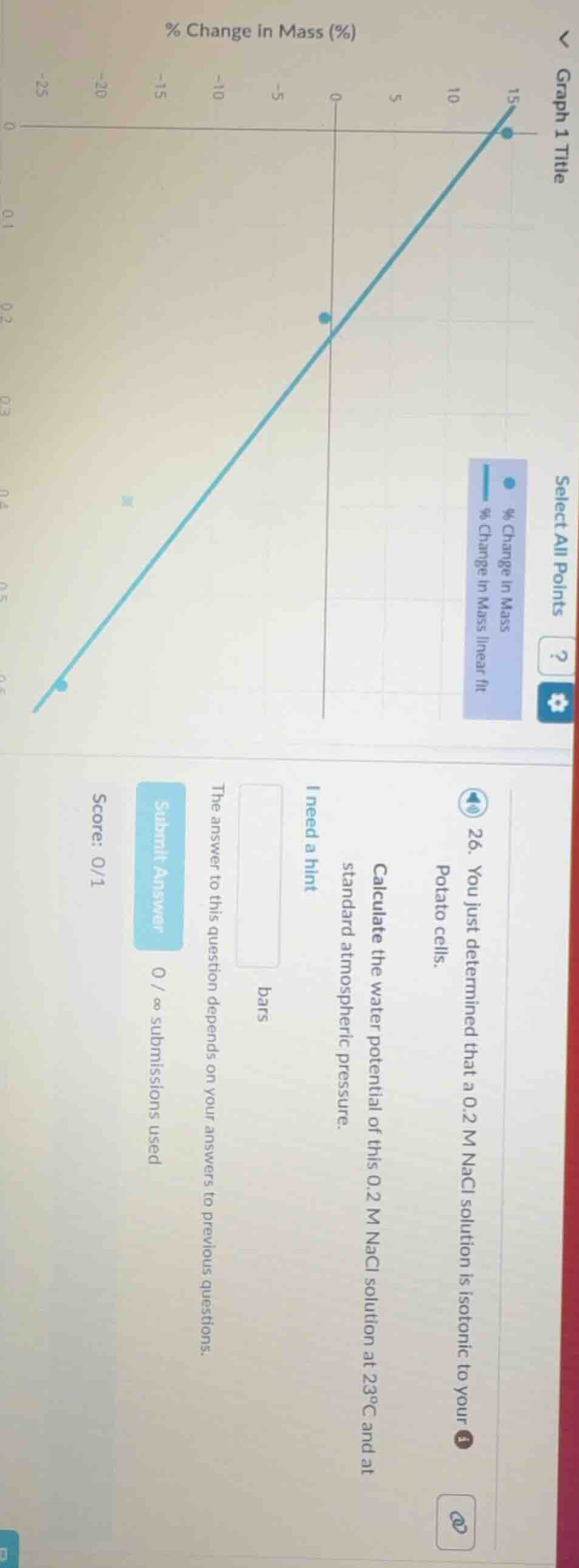
- you just determined that a 0.2 m nacl solution is isotonic to your potato cells. calculate the water potential of this 0.2 m nacl solution at 23°c and at standard atmospheric pressure. bars the answer to this question depends on your answers to previous questions. submit answer 0 / ∞ submissions used score: 0/1
Step1: Recall water potential formula
For a solution at atmospheric pressure, water potential $\Psi = \Psi_s$ (solute potential), and $\Psi_s = -iCRT$
Step2: Define variables
$i=2$ (NaCl dissociates into 2 ions), $C=0.2\ \text{M}$, $R=0.0831\ \text{L·bar/(mol·K)}$, $T=23+273=296\ \text{K}$
Step3: Calculate solute potential
$\Psi_s = -2 \times 0.2 \times 0.0831 \times 296$
$\Psi_s = -2 \times 0.2 \times 24.5976$
$\Psi_s = -9.83904$
Step4: Relate to water potential
At standard atmospheric pressure, pressure potential $\Psi_p=0$, so $\Psi = \Psi_s$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
-9.84 bars (or ~-9.8 bars)