QUESTION IMAGE
Question
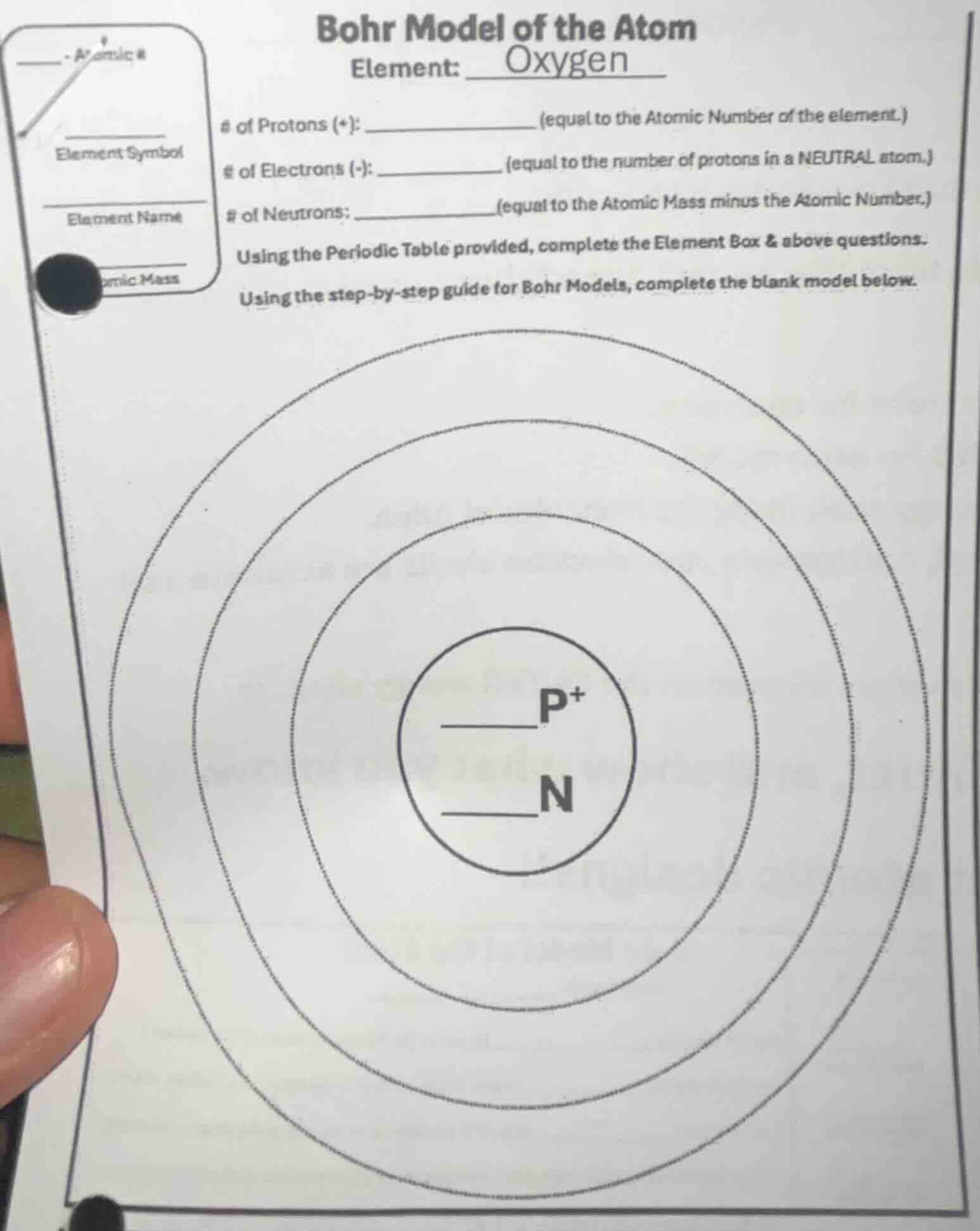
bohr model of the atom
element: oxygen
of protons (+): ______________ (equal to the atomic number of the element.)
of electrons (-): ____________ (equal to the number of protons in a neutral atom.)
of neutrons: ______________ (equal to the atomic mass minus the atomic number.)
using the periodic table provided, complete the element box & above questions.
using the step - by - step guide for bohr models, complete the blank model below.
bohr model diagram with circles for electron shells, and a central circle with p⁺ (protons) and n (neutrons) labels, with blanks to fill in the number of protons and neutrons, and electron shells to fill with electrons
Step1: Find Oxygen's atomic number
From the periodic table, oxygen (O) has an atomic number of 8. This equals the number of protons.
Step2: Determine electron count
For a neutral atom, electrons = protons = 8.
Step3: Calculate neutron count
Oxygen's atomic mass is ~16. Neutrons = Atomic Mass - Atomic Number = $16 - 8 = 8$.
Step4: Fill electron shells
Bohr model rules: 1st shell holds max 2 electrons, 2nd shell holds max 8. For oxygen: 1st shell = 2 electrons, remaining $8-2=6$ electrons in 2nd shell.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Element Box:
- Atomic #: 8
- Element Symbol: O
- Element Name: Oxygen
- Atomic Mass: 16
Particle Counts:
- # of Protons (+): 8
- # of Electrons (-): 8
- # of Neutrons: 8
Bohr Model Details:
- Nucleus: 8 P⁺, 8 N
- 1st innermost electron shell: 2 electrons
- 2nd electron shell: 6 electrons
(The outermost third shell remains empty as oxygen only uses the first two shells for its electrons)