QUESTION IMAGE
Question
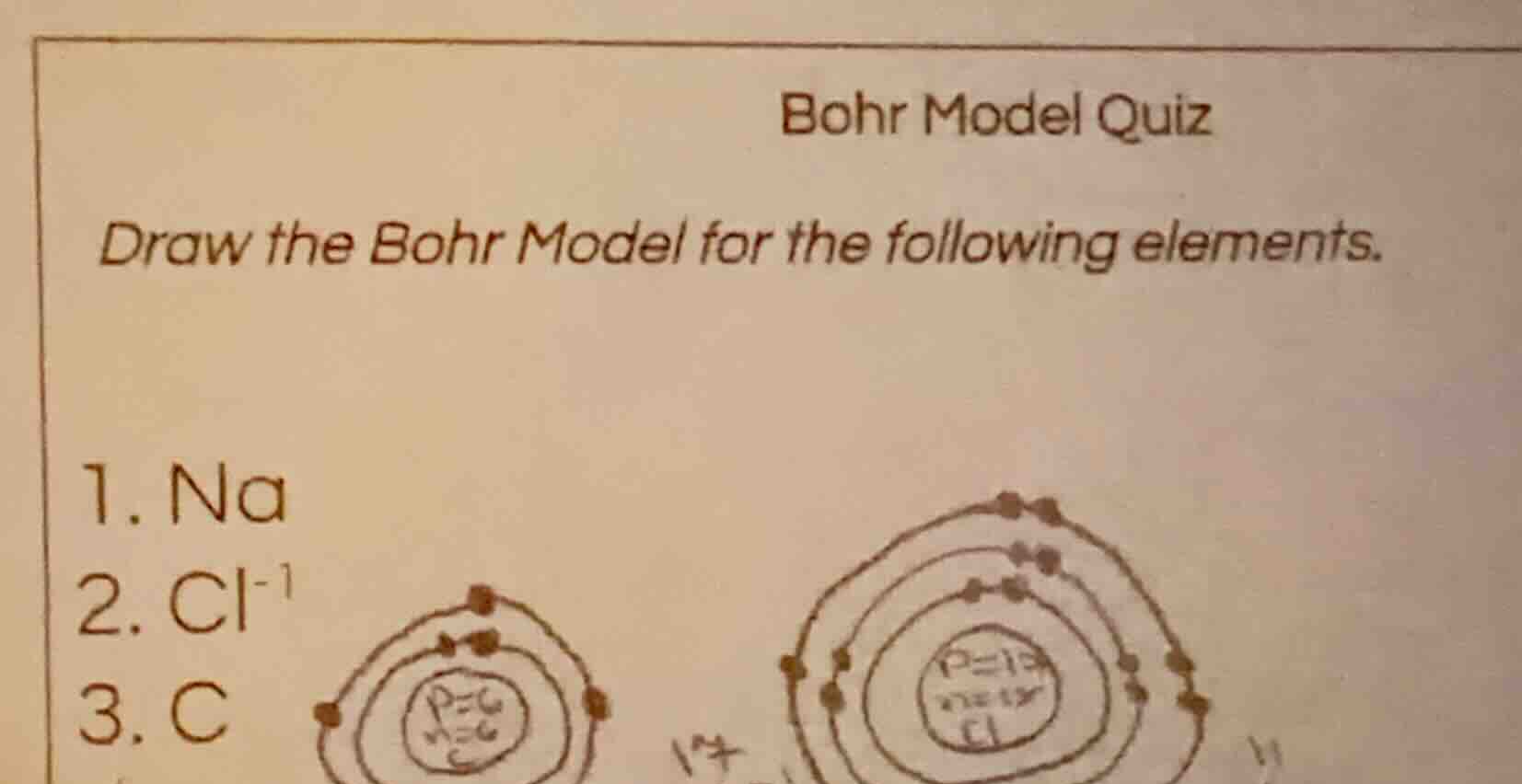
bohr model quiz
draw the bohr model for the following elements.
- na
- cl⁻¹
- c
Step1: Find Na's atomic number
Sodium (Na) has atomic number $Z=11$, so protons $=11$, electrons $=11$. Electron shell capacity: $2, 8, 1$.
Step2: Find Cl⁻'s electron count
Chlorine (Cl) has $Z=17$, neutral electrons $=17$. $Cl^-$ gains 1 electron, so electrons $=18$. Shells: $2, 8, 8$.
Step3: Find C's atomic number
Carbon (C) has $Z=6$, protons $=6$, electrons $=6$. Shells: $2, 4$.
Step4: Define Bohr Model structure
Each model has a nucleus labeled with protons ($p$) and neutrons ($n$, approximate mass number - protons: Na mass=23, $n=12$; Cl mass=35, $n=18$; C mass=12, $n=6$), plus electron shells with correct electron counts.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- Sodium (Na) Bohr Model:
- Nucleus: Label with $p=11$, $n=12$
- 1st inner shell: 2 electrons
- 2nd shell: 8 electrons
- 3rd outer shell: 1 electron
- Chloride Ion (Cl⁻) Bohr Model:
- Nucleus: Label with $p=17$, $n=18$
- 1st inner shell: 2 electrons
- 2nd shell: 8 electrons
- 3rd outer shell: 8 electrons
- Carbon (C) Bohr Model:
- Nucleus: Label with $p=6$, $n=6$
- 1st inner shell: 2 electrons
- 2nd outer shell: 4 electrons
(Visual structure: Concentric circles around the nucleus, with dots representing electrons on each circle matching the counts above)