QUESTION IMAGE
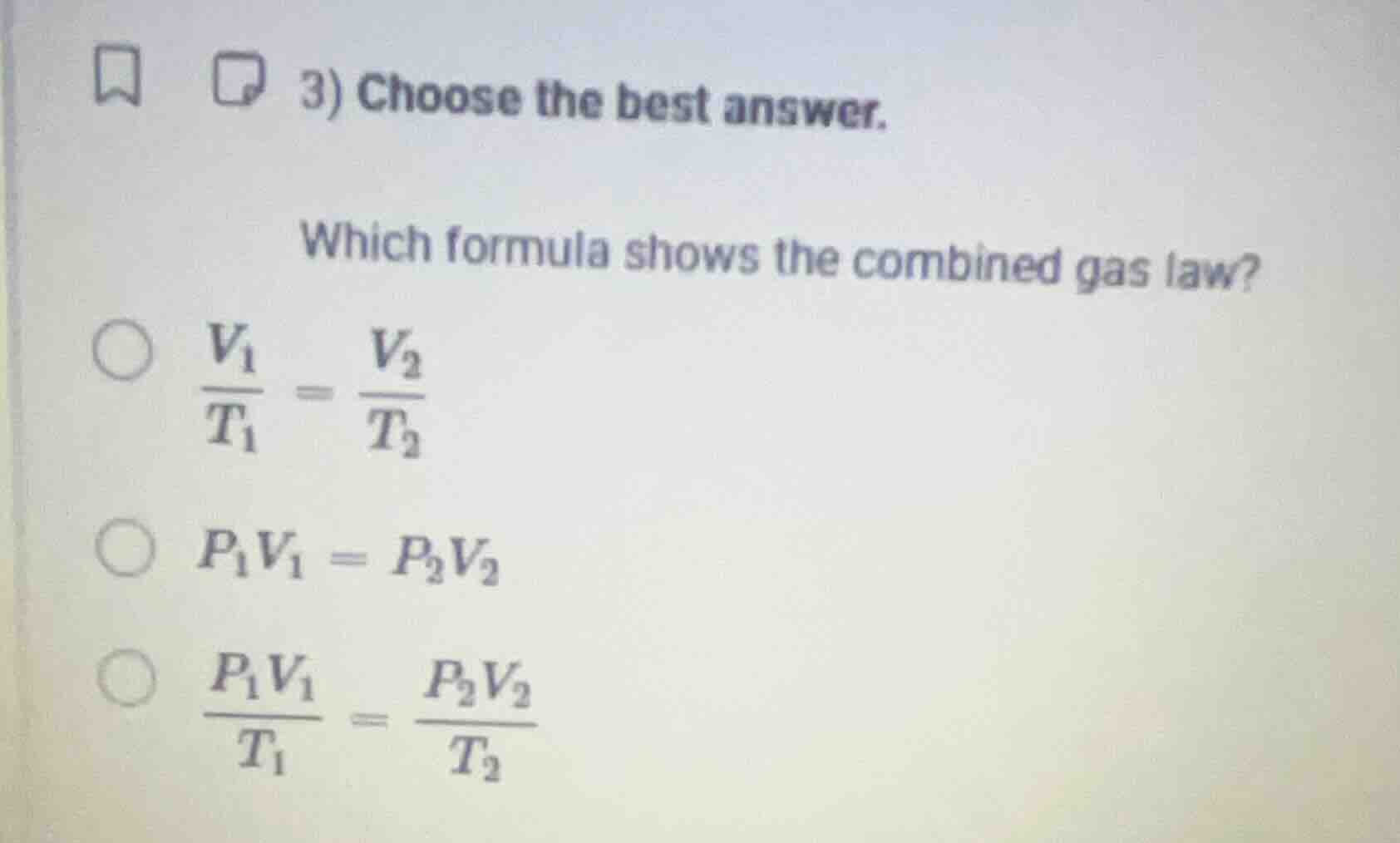
Question
- choose the best answer.
which formula shows the combined gas law?
$\frac{v_1}{t_1} = \frac{v_2}{t_2}$
$p_1v_1 = p_2v_2$
$\frac{p_1v_1}{t_1} = \frac{p_2v_2}{t_2}$
Brief Explanations
The combined gas law merges Boyle's Law, Charles's Law, and Gay-Lussac's Law, relating pressure (P), volume (V), and absolute temperature (T) of a fixed amount of gas. The first option is Charles's Law (constant pressure), the second is Boyle's Law (constant temperature), while the third combines all three variables.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
$\boldsymbol{\frac{P_1V_1}{T_1} = \frac{P_2V_2}{T_2}}$