QUESTION IMAGE
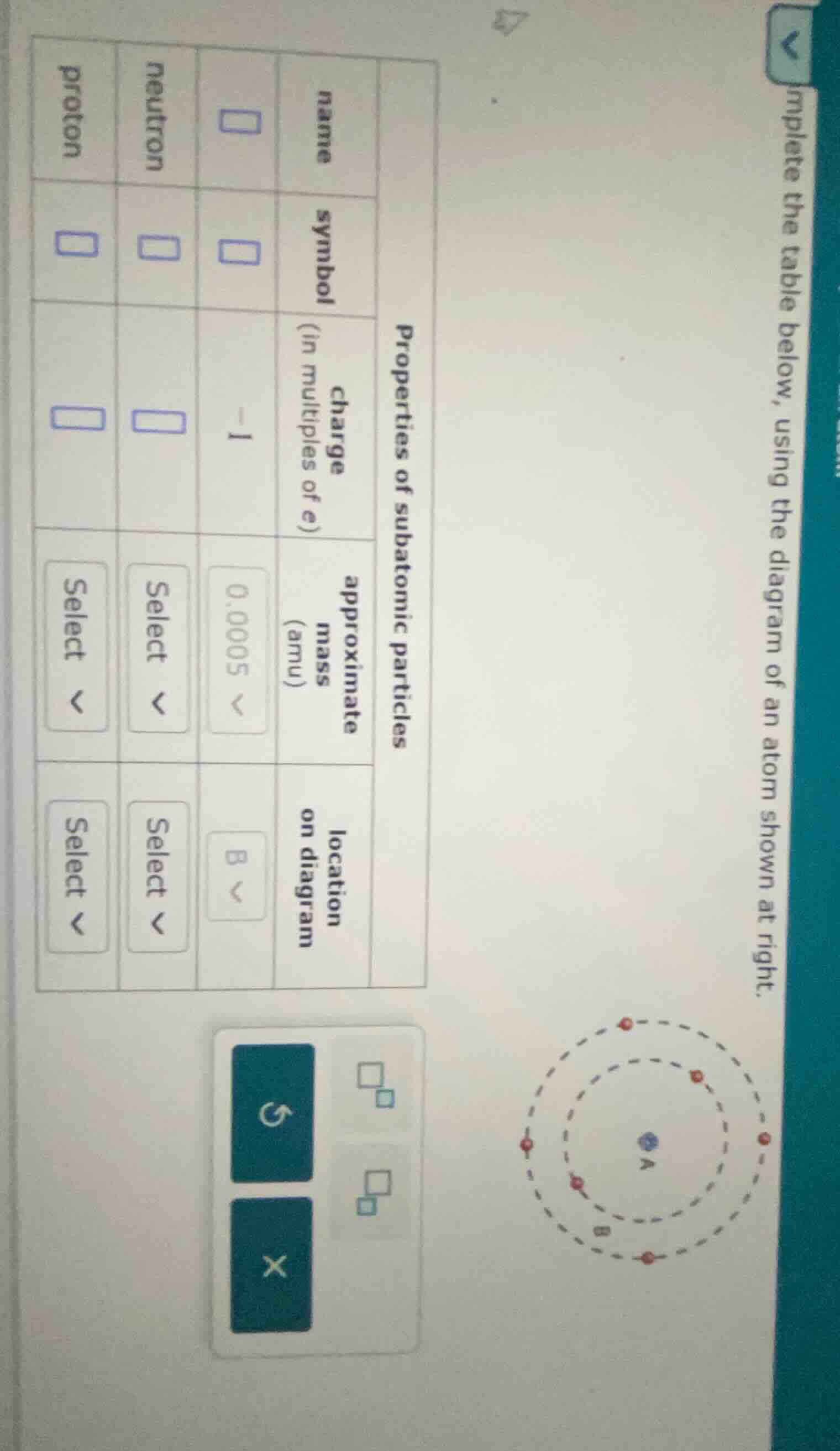
Question
complete the table below, using the diagram of an atom shown at right.
properties of subatomic particles
| name | symbol | charge (in multiples of e) | approximate mass (amu) | location on diagram | |
|---|---|---|---|---|---|
| proton | select | select | |||
| neutron | select | select |
Step1: Identify the particle with charge -1
The particle with charge -1 (in multiples of \( e \)) is the electron. So the first row (name) should be "electron".
Step2: Determine the symbol of electron
The symbol for electron is \( e^- \) (or sometimes just \( e \), but in the context of subatomic particles, \( e^- \) is common).
Step3: Determine the charge of neutron and proton
Neutron has a charge of 0 (in multiples of \( e \)), and proton has a charge of +1 (in multiples of \( e \)).
Step4: Determine the approximate mass of neutron and proton
The approximate mass of neutron is 1 amu, and the approximate mass of proton is also 1 amu (electron is ~0.0005 amu as given).
Step5: Determine the location of particles
In the atom diagram, "A" is the nucleus (where protons and neutrons are located), and "B" is the electron cloud (where electrons are located). So electron is at location B, proton and neutron are at location A.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
| name | symbol | charge (in multiples of \( e \)) | approximate mass (amu) | location on diagram |
|---|---|---|---|---|
| neutron | \( n \) | 0 | 1 | A |
| proton | \( p^+ \) | +1 | 1 | A |