QUESTION IMAGE
Question
select the atomic models that belong to the same element.
To determine which atomic models belong to the same element, we use the fact that the number of protons (atomic number) defines an element. So we check the number of protons (red 'P' particles) in each nucleus and the number of electrons (blue particles, equal to protons in neutral atoms).
Step1: Analyze each model
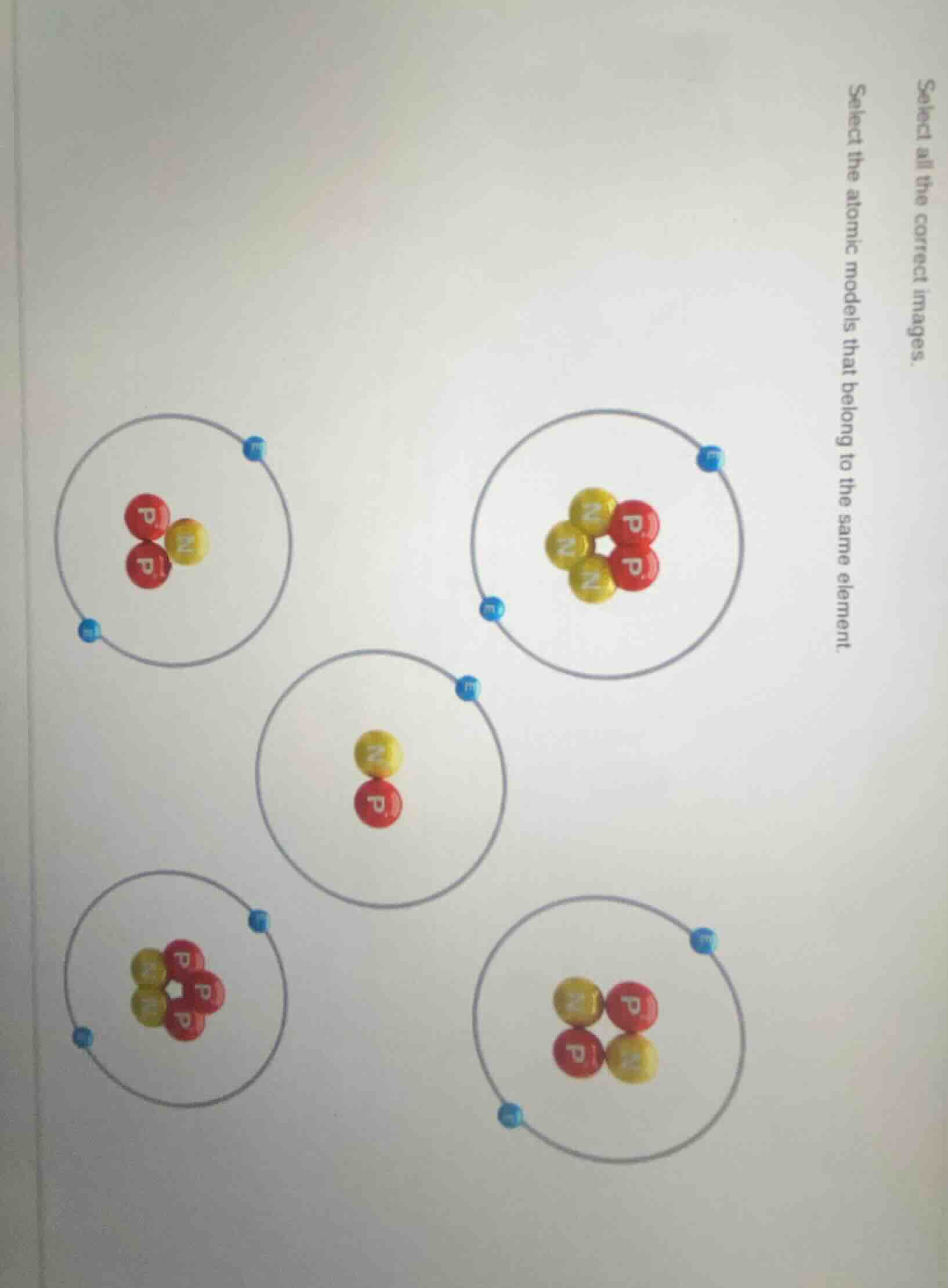
- Top - left: Protons (P) = 2, Neutrons (N) = 1, Electrons (e) = 2.
- Top - right: Protons (P) = 2, Neutrons (N) = 3, Electrons (e) = 2.
- Middle: Protons (P) = 1, Neutrons (N) = 1, Electrons (e) = 1.
- Bottom - left: Protons (P) = 3, Neutrons (N) = 2, Electrons (e) = 2 (wait, no—wait, electrons should equal protons. Wait, bottom - left: electrons = 2, protons = 3? No, maybe I miscounted. Wait, let's re - check:
- Top - left: 2 P, 1 N, 2 e.
- Top - right: 2 P, 3 N, 2 e.
- Middle: 1 P, 1 N, 1 e.
- Bottom - left: 3 P, 2 N, 2 e (incorrect, electrons should equal protons. Maybe a typo? Wait, no—wait the bottom - right: 2 P, 2 N, 2 e. Wait, no, let's list protons (P) count:
Top - left: P = 2
Top - right: P = 2
Middle: P = 1
Bottom - left: P = 3
Bottom - right: P = 2 (wait, bottom - right: two red P, two yellow N, two blue e. So P = 2, N = 2, e = 2.
Wait, so the models with P = 2 are top - left (2P,1N,2e), top - right (2P,3N,2e), and bottom - right (2P,2N,2e). Wait, but let's check electrons: in neutral atoms, electrons = protons. So:
- Top - left: e = 2, P = 2 → neutral.
- Top - right: e = 2, P = 2 → neutral.
- Middle: e = 1, P = 1 → neutral.
- Bottom - left: e = 2, P = 3 → ion (but the question is about same element, which is defined by P. So elements with same P (atomic number) are same element, regardless of neutrons (isotopes) or charge (but here most are neutral).
So the models with P = 2 are top - left, top - right, and bottom - right? Wait, no, bottom - right: two P, two N, two e. Top - left: two P, one N, two e. Top - right: two P, three N, two e. So these three have P = 2, so same element (atomic number 2, like Helium isotopes and neutral Helium). The middle has P = 1 (Hydrogen), bottom - left has P = 3 (Lithium).
Wait, maybe I mis - saw the bottom - left. Let's re - examine the bottom - left: the nucleus has three red P and two yellow N, and two blue e. So P = 3, e = 2 → cation (Li⁺), but element is Lithium (P = 3). The middle is P = 1 (Hydrogen), top - left, top - right, bottom - right: P = 2 (Helium - like, with different neutrons: 1, 2, 3 neutrons? Wait top - left: N = 1, top - right: N = 3, bottom - right: N = 2. So these are isotopes of the same element (atomic number 2).
So the atomic models with 2 protons (same atomic number) are top - left, top - right, and bottom - right? Wait, but let's check the original image again (as per the user's image:
Top - left: 2 P, 1 N, 2 e
Top - right: 2 P, 3 N, 2 e
Middle: 1 P, 1 N, 1 e
Bottom - left: 3 P, 2 N, 2 e
Bottom - right: 2 P, 2 N, 2 e
So the ones with P = 2 (atomic number 2) are top - left, top - right, and bottom - right. But maybe the bottom - left is a mistake, or maybe I miscounted. Alternatively, maybe the correct ones are top - left, top - right, and bottom - right? Wait, no, let's check the electron count again. In a neutral atom, electrons = protons. So:
- Top - left: e = 2, P = 2 → neutral.
- Top - right: e = 2, P = 2 → neutral.
- Middle: e = 1, P = 1 → neutral.
- Bottom - left: e = 2, P = 3 → Li⁺ (element Li, P = 3).
- Bottom - right: e = 2, P = 2 → neutral (He).
So the same element (P = 2) are top - left, top - right, and bottom - right? Wait, but maybe the bottom - right is also 2P. So the answer…
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
The atomic models that belong to the same element are the top - left, top - right, and bottom - right models (assuming the bottom - right has 2 protons, 2 neutrons, and 2 electrons; top - left has 2 protons, 1 neutron, and 2 electrons; top - right has 2 protons, 3 neutrons, and 2 electrons). If we consider the visual:
- Top - left (2P,1N,2e)
- Top - right (2P,3N,2e)
- Bottom - right (2P,2N,2e)
These have the same number of protons (atomic number 2), so they are the same element.