QUESTION IMAGE
Question
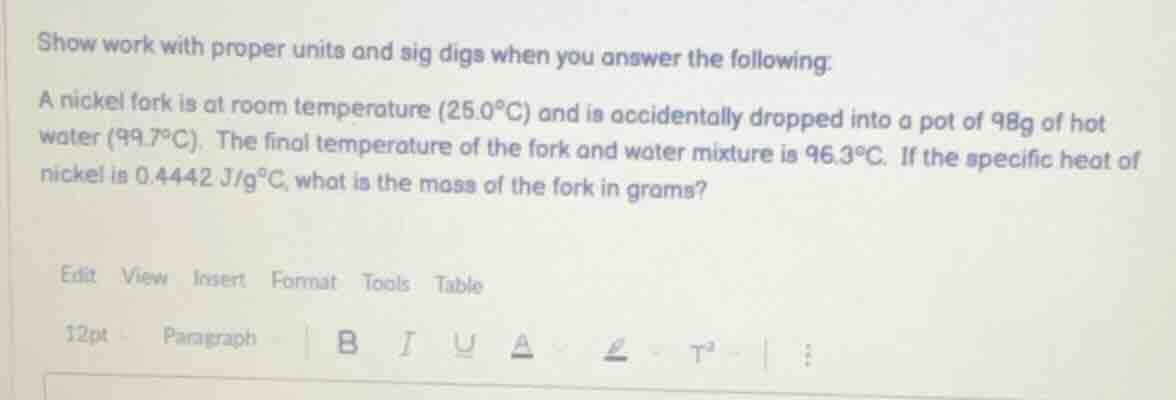
show work with proper units and sig digs when you answer the following: a nickel fork is at room temperature (25.0°c) and is accidentally dropped into a pot of 98g of hot water (99.7°c). the final temperature of the fork and water mixture is 96.3°c. if the specific heat of nickel is 0.4442 j/g°c, what is the mass of the fork in grams?
Step1: Set heat balance equation
Heat lost by water = Heat gained by fork:
$$m_w c_w \Delta T_w = m_n c_n \Delta T_n$$
(where $m_w$ = mass of water, $c_w=4.184\ \text{J/g}^\circ\text{C}$ (specific heat of water), $\Delta T_w$ = temp change of water; $m_n$ = mass of nickel fork, $c_n=0.4442\ \text{J/g}^\circ\text{C}$, $\Delta T_n$ = temp change of nickel)
Step2: Calculate temperature changes
$\Delta T_w = 99.7^\circ\text{C} - 96.3^\circ\text{C} = 3.4^\circ\text{C}$
$\Delta T_n = 96.3^\circ\text{C} - 25.0^\circ\text{C} = 71.3^\circ\text{C}$
Step3: Rearrange for $m_n$
$$m_n = \frac{m_w c_w \Delta T_w}{c_n \Delta T_n}$$
Step4: Substitute values
$$m_n = \frac{98\ \text{g} \times 4.184\ \text{J/g}^\circ\text{C} \times 3.4^\circ\text{C}}{0.4442\ \text{J/g}^\circ\text{C} \times 71.3^\circ\text{C}}$$
$$m_n = \frac{98 \times 4.184 \times 3.4}{0.4442 \times 71.3}\ \text{g}$$
$$m_n = \frac{1390.1888}{31.67146}\ \text{g} \approx 44\ \text{g}$$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
$\boldsymbol{44\ \text{grams}}$