QUESTION IMAGE
Question
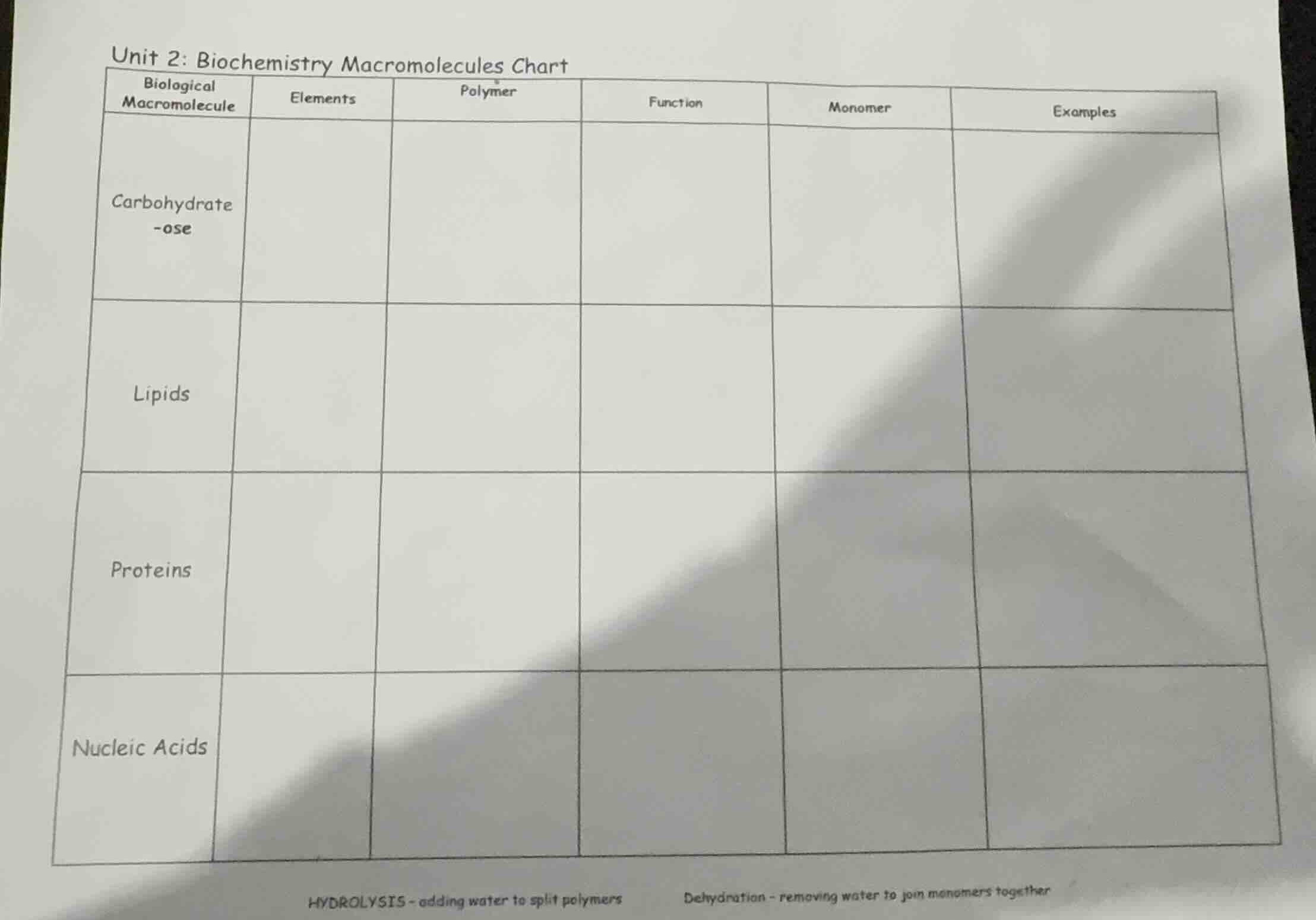
unit 2: biochemistry macromolecules chart
biological macromolecule | elements | polymer | function | monomer | examples
carbohydrate -ose
lipids
proteins
nucleic acids
hydrolysis - adding water to split polymers dehydration - removing water to join monomers together
To complete the "Biochemistry Macromolecules Chart", we analyze each macromolecule:
1. Carbohydrate (-ose)
- Elements: $C, H, O$ (ratio $1:2:1$, e.g., $C_6H_{12}O_6$)
- Polymer: Polysaccharide (e.g., starch, glycogen, cellulose)
- Function: Primary energy source (short-term energy); structural support (e.g., cellulose in plants).
- Monomer: Monosaccharide (e.g., glucose, fructose, galactose)
- Examples: Glucose, sucrose, starch, cellulose
2. Lipids
- Elements: $C, H, O$ (often more $H$ than $O$; some contain $P, N$)
- Polymer: No true polymer (lipids are non-polar and do not form long chains like others)
- Function: Long-term energy storage; insulation; cell membrane structure (phospholipids); hormone signaling (steroids).
- Monomer: Fatty acids + glycerol (triglycerides: 3 fatty acids + 1 glycerol)
- Examples: Fats, oils, phospholipids, cholesterol
3. Proteins
- Elements: $C, H, O, N$ (sometimes $S$)
- Polymer: Polypeptide (chain of amino acids linked by peptide bonds)
- Function: Enzymes (catalyze reactions); structural (e.g., collagen); transport (e.g., hemoglobin); immune (e.g., antibodies); signaling (e.g., hormones).
- Monomer: Amino acid (20 different types, each with $-NH_2$, $-COOH$, and a unique $R$-group)
- Examples: Hemoglobin, insulin, keratin, enzymes (e.g., amylase)
4. Nucleic Acids
- Elements: $C, H, O, N, P$
- Polymer: Polynucleotide (chain of nucleotides)
- Function: Store and transmit genetic information (DNA: genetic code; RNA: protein synthesis).
- Monomer: Nucleotide (3 parts: sugar, phosphate, nitrogenous base)
- Examples: DNA (deoxyribonucleic acid), RNA (ribonucleic acid)
Filling the Chart (Table Format)
| Biological Macromolecule | Elements | Polymer | Function | Monomer | Examples |
|---|---|---|---|---|---|
| Lipids | $C, H, O$ (±P,N) | No true polymer | Energy storage; insulation; membrane structure | Fatty acids + glycerol | Fats, phospholipids, cholesterol |
| Proteins | $C, H, O, N$ (±S) | Polypeptide | Enzymes, structure, transport, immunity | Amino acid | Hemoglobin, insulin, enzymes |
| Nucleic Acids | $C, H, O, N, P$ | Polynucleotide | Genetic information storage/transmission | Nucleotide | DNA, RNA |
Key Processes (Bottom of Chart)
- Hydrolysis: Breaks polymers into monomers by adding water (e.g., digesting starch into glucose).
- Dehydration Synthesis: Joins monomers into polymers by removing water (e.g., glucose → starch).
If you need to summarize or format this differently, let me know!
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
To complete the "Biochemistry Macromolecules Chart", we analyze each macromolecule:
1. Carbohydrate (-ose)
- Elements: $C, H, O$ (ratio $1:2:1$, e.g., $C_6H_{12}O_6$)
- Polymer: Polysaccharide (e.g., starch, glycogen, cellulose)
- Function: Primary energy source (short-term energy); structural support (e.g., cellulose in plants).
- Monomer: Monosaccharide (e.g., glucose, fructose, galactose)
- Examples: Glucose, sucrose, starch, cellulose
2. Lipids
- Elements: $C, H, O$ (often more $H$ than $O$; some contain $P, N$)
- Polymer: No true polymer (lipids are non-polar and do not form long chains like others)
- Function: Long-term energy storage; insulation; cell membrane structure (phospholipids); hormone signaling (steroids).
- Monomer: Fatty acids + glycerol (triglycerides: 3 fatty acids + 1 glycerol)
- Examples: Fats, oils, phospholipids, cholesterol
3. Proteins
- Elements: $C, H, O, N$ (sometimes $S$)
- Polymer: Polypeptide (chain of amino acids linked by peptide bonds)
- Function: Enzymes (catalyze reactions); structural (e.g., collagen); transport (e.g., hemoglobin); immune (e.g., antibodies); signaling (e.g., hormones).
- Monomer: Amino acid (20 different types, each with $-NH_2$, $-COOH$, and a unique $R$-group)
- Examples: Hemoglobin, insulin, keratin, enzymes (e.g., amylase)
4. Nucleic Acids
- Elements: $C, H, O, N, P$
- Polymer: Polynucleotide (chain of nucleotides)
- Function: Store and transmit genetic information (DNA: genetic code; RNA: protein synthesis).
- Monomer: Nucleotide (3 parts: sugar, phosphate, nitrogenous base)
- Examples: DNA (deoxyribonucleic acid), RNA (ribonucleic acid)
Filling the Chart (Table Format)
| Biological Macromolecule | Elements | Polymer | Function | Monomer | Examples |
|---|---|---|---|---|---|
| Lipids | $C, H, O$ (±P,N) | No true polymer | Energy storage; insulation; membrane structure | Fatty acids + glycerol | Fats, phospholipids, cholesterol |
| Proteins | $C, H, O, N$ (±S) | Polypeptide | Enzymes, structure, transport, immunity | Amino acid | Hemoglobin, insulin, enzymes |
| Nucleic Acids | $C, H, O, N, P$ | Polynucleotide | Genetic information storage/transmission | Nucleotide | DNA, RNA |
Key Processes (Bottom of Chart)
- Hydrolysis: Breaks polymers into monomers by adding water (e.g., digesting starch into glucose).
- Dehydration Synthesis: Joins monomers into polymers by removing water (e.g., glucose → starch).
If you need to summarize or format this differently, let me know!