QUESTION IMAGE
Question
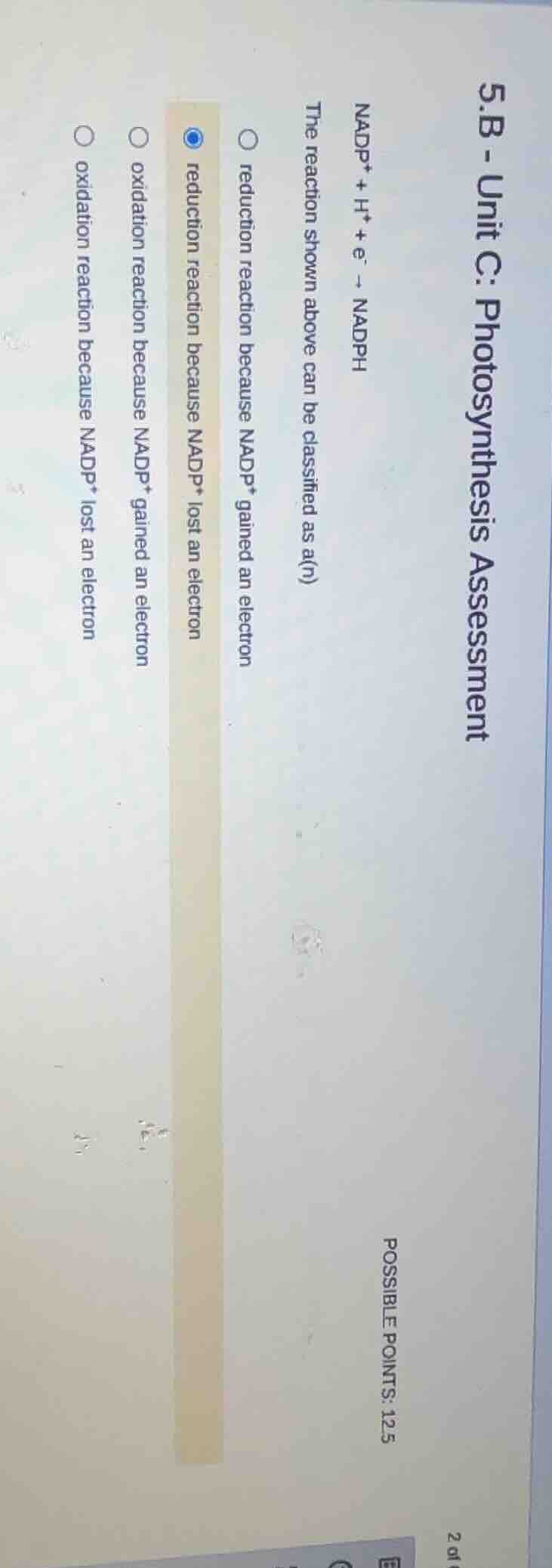
5.b - unit c: photosynthesis assessment\
nadp⁺ + h⁺ + e⁻ → nadph\
the reaction shown above can be classified as a(n)\
\
\\u25ef reduction reaction because nadp⁺ gained an electron\
\\u25cf reduction reaction because nadp⁺ lost an electron\
\\u25ef oxidation reaction because nadp⁺ gained an electron\
\\u25ef oxidation reaction because nadp⁺ lost an electron
In the reaction \( \text{NADP}^+ + \text{H}^+ + \text{e}^-
ightarrow \text{NADPH} \), \( \text{NADP}^+ \) gains an electron (along with a proton). A reduction reaction is defined as a gain of electrons (or a decrease in oxidation state). So this is a reduction reaction because \( \text{NADP}^+ \) gained an electron.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
A. reduction reaction because \( \text{NADP}^+ \) gained an electron