QUESTION IMAGE
Question
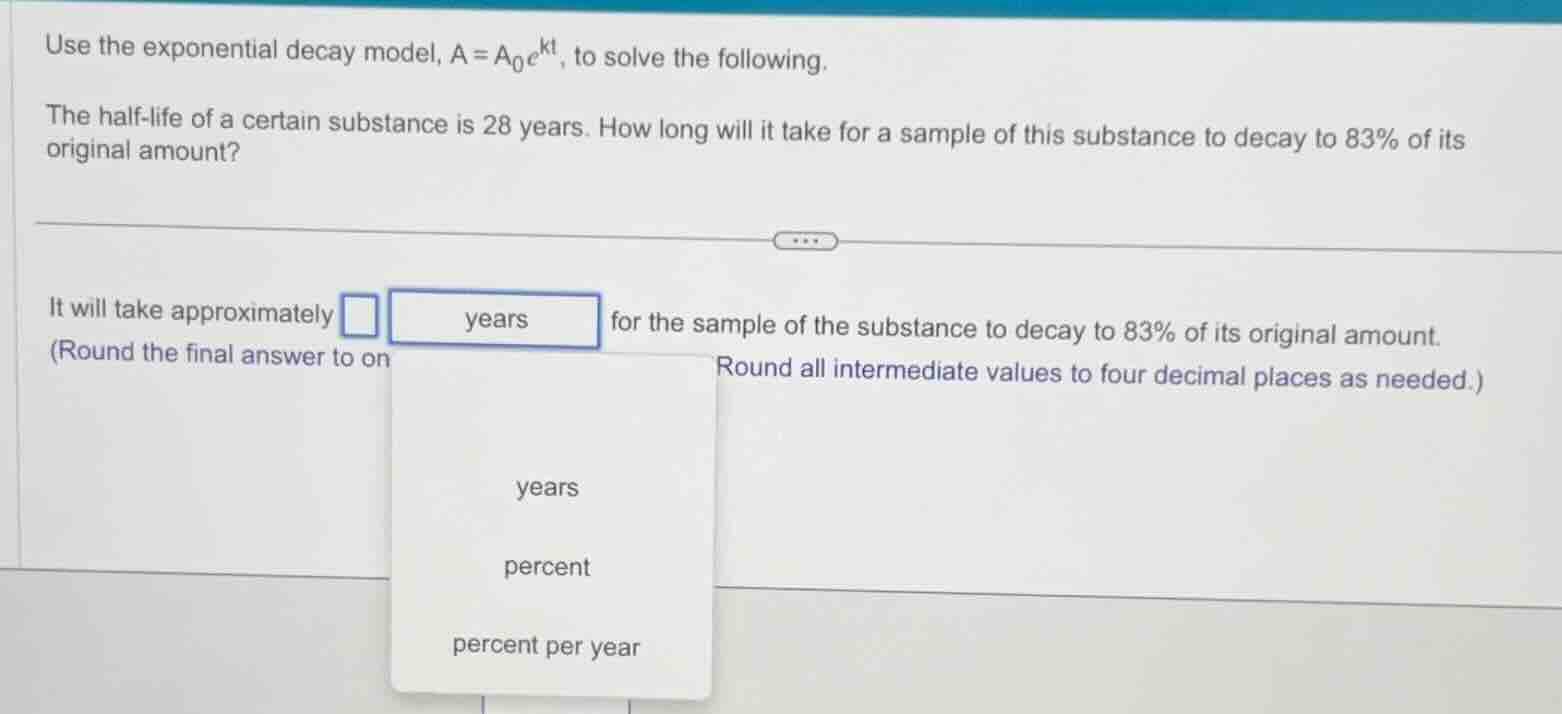
use the exponential decay model, $a = a_0e^{kt}$, to solve the following. the half - life of a certain substance is 28 years. how long will it take for a sample of this substance to decay to 83% of its original amount? it will take approximately years for the sample of the substance to decay to 83% of its original amount. (round the final answer to on round all intermediate values to four decimal places as needed.) years percent percent per year
Step1: Find the decay constant \( k \)
We know that for half - life, when \( t = 28 \) years, \( A=\frac{1}{2}A_0 \). Substitute into the exponential decay model \( A = A_0e^{kt} \):
\(\frac{1}{2}A_0=A_0e^{k\times28}\)
Divide both sides by \( A_0 \) (since \( A_0
eq0 \)): \(\frac{1}{2}=e^{28k}\)
Take the natural logarithm of both sides: \(\ln(\frac{1}{2})=\ln(e^{28k})\)
Using the property \(\ln(e^x)=x\), we get: \(\ln(\frac{1}{2}) = 28k\)
So, \( k=\frac{\ln(\frac{1}{2})}{28}=\frac{-\ln(2)}{28}\approx\frac{- 0.6931}{28}\approx - 0.02475\)
Step2: Find the time \( t \) when \( A = 0.83A_0 \)
Substitute \( A = 0.83A_0 \) and \( k\approx - 0.02475 \) into \( A = A_0e^{kt} \):
\(0.83A_0=A_0e^{-0.02475t}\)
Divide both sides by \( A_0 \): \(0.83 = e^{-0.02475t}\)
Take the natural logarithm of both sides: \(\ln(0.83)=\ln(e^{-0.02475t})\)
Using the property \(\ln(e^x)=x\), we have: \(\ln(0.83)=- 0.02475t\)
Then, \( t=\frac{\ln(0.83)}{- 0.02475}\)
We know that \(\ln(0.83)\approx - 0.1863\)
So, \( t=\frac{-0.1863}{-0.02475}\approx7.53\)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
7.53