QUESTION IMAGE
Question
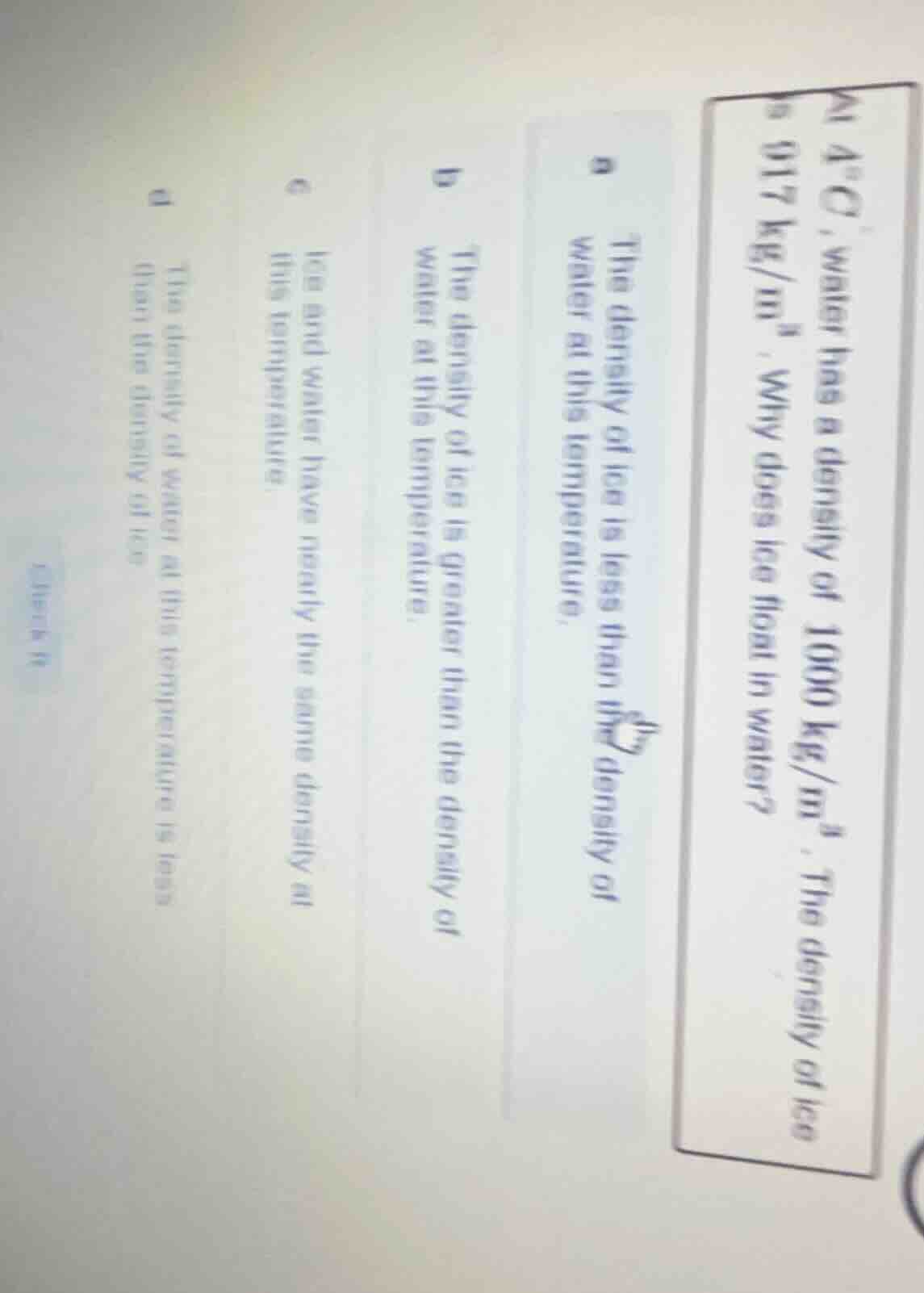
at 4°c, water has a density of 1000 kg/m³. the density of ice is 917 kg/m³. why does ice float in water?
a the density of ice is less than the density of water at this temperature.
b the density of ice is greater than the density of water at this temperature.
c ice and water have nearly the same density at this temperature.
d the density of water at this temperature is less than the density of ice.
To determine why ice floats in water, we use the principle of buoyancy and the concept of density. An object floats in a fluid if its density is less than the density of the fluid. Given:
- Density of water ($
ho_{water}$) = $1000\space kg/m^3$
- Density of ice ($
ho_{ice}$) = $917\space kg/m^3$
Since $
ho_{ice} = 917\space kg/m^3$ is less than $
ho_{water} = 1000\space kg/m^3$, ice is less dense than water. According to Archimedes' principle, when an object is placed in a fluid, the buoyant force acting on it is equal to the weight of the fluid displaced. If the object's density is less than the fluid's density, the buoyant force (which depends on the fluid's density) is greater than the object's weight, causing it to float.
Now let's analyze the options:
- Option a: States "The density of ice is less than the density of water at this temperature." This matches our reasoning as $
ho_{ice} (917\space kg/m^3) <
ho_{water} (1000\space kg/m^3)$.
- Option b: Says the density of ice is greater than water, which is false.
- Option c: Claims ice and water have nearly the same density, which is false as their densities are significantly different (917 vs 1000 $kg/m^3$).
- Option d: States the density of water at this temperature is less than the density of ice, which is false (water is more dense than ice here).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
a. The density of ice is less than the density of water at this temperature.