QUESTION IMAGE
Question
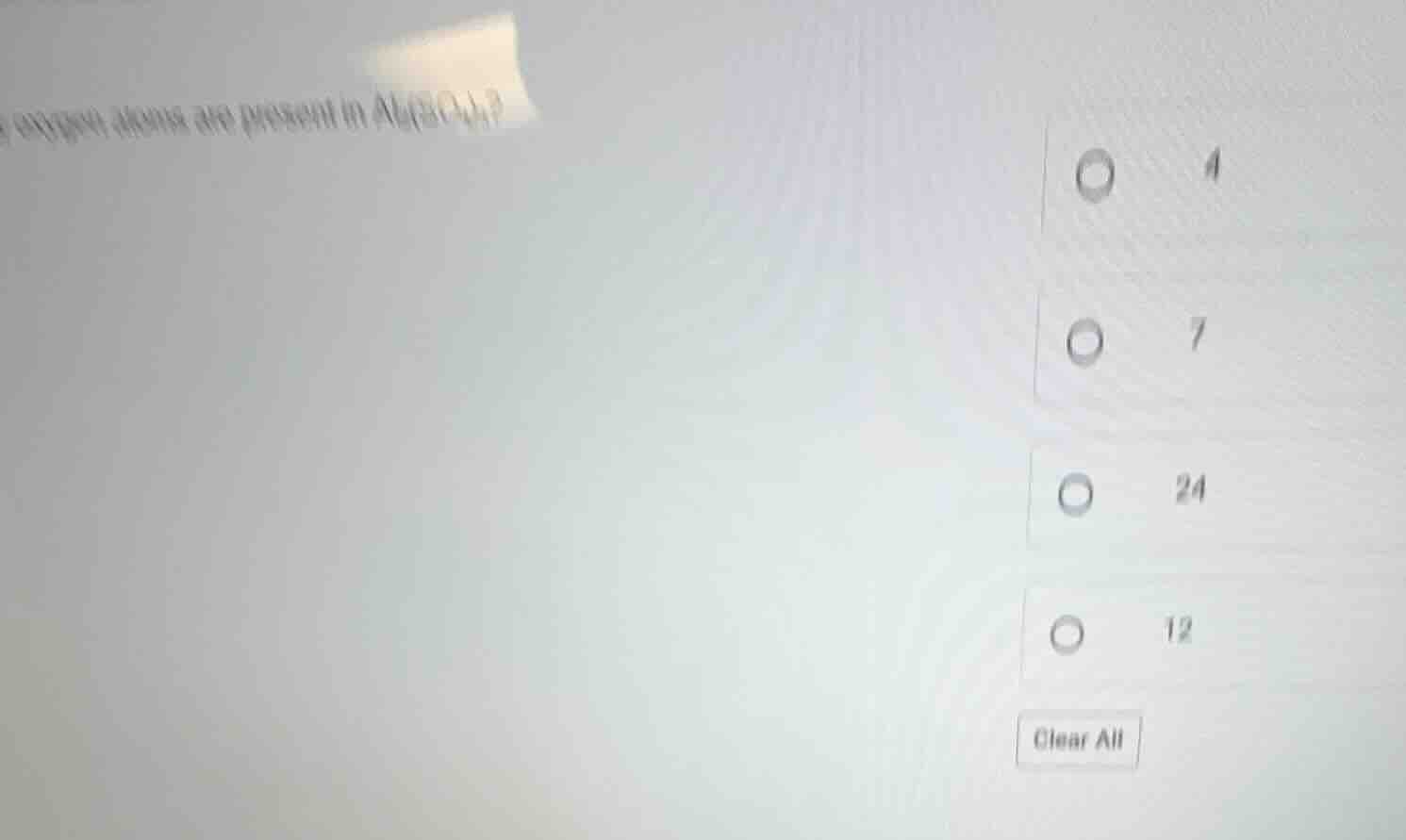
what atoms are present in al₂(so₄)₃? (options: 4, 7, 24, 12)
Step1: Identify atoms in \( \text{Al}_2(\text{SO}_4)_3 \)
- Aluminum (\( \text{Al} \)): subscript 2, so 2 atoms.
- Sulfur (\( \text{S} \)): in \( \text{SO}_4 \), subscript 1, and \( (\text{SO}_4)_3 \), so \( 1\times3 = 3 \) atoms.
- Oxygen (\( \text{O} \)): in \( \text{SO}_4 \), subscript 4, and \( (\text{SO}_4)_3 \), so \( 4\times3 = 12 \) atoms.
Step2: Sum the number of atoms
Total atoms \( = 2 + 3 + 12 = 17 \)? Wait, no, maybe the question is about "different atoms" (elements) present? Wait, the options are 4,7,24,12. Wait, maybe the formula is \( \text{Al}_2(\text{SO}_4)_3 \), the elements are Al, S, O. Wait, no, maybe the question is about the number of oxygen atoms? Wait, \( \text{SO}_4 \) has 4 O, times 3 is 12. Oh! The options include 12. So maybe the question is "How many oxygen atoms are present in \( \text{Al}_2(\text{SO}_4)_3 \)?"
So:
- In \( \text{SO}_4^{2-} \), O atoms: 4.
- There are 3 \( \text{SO}_4^{2-} \) groups, so \( 4\times3 = 12 \).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
12 (corresponding to the option with 12)