QUESTION IMAGE
Question
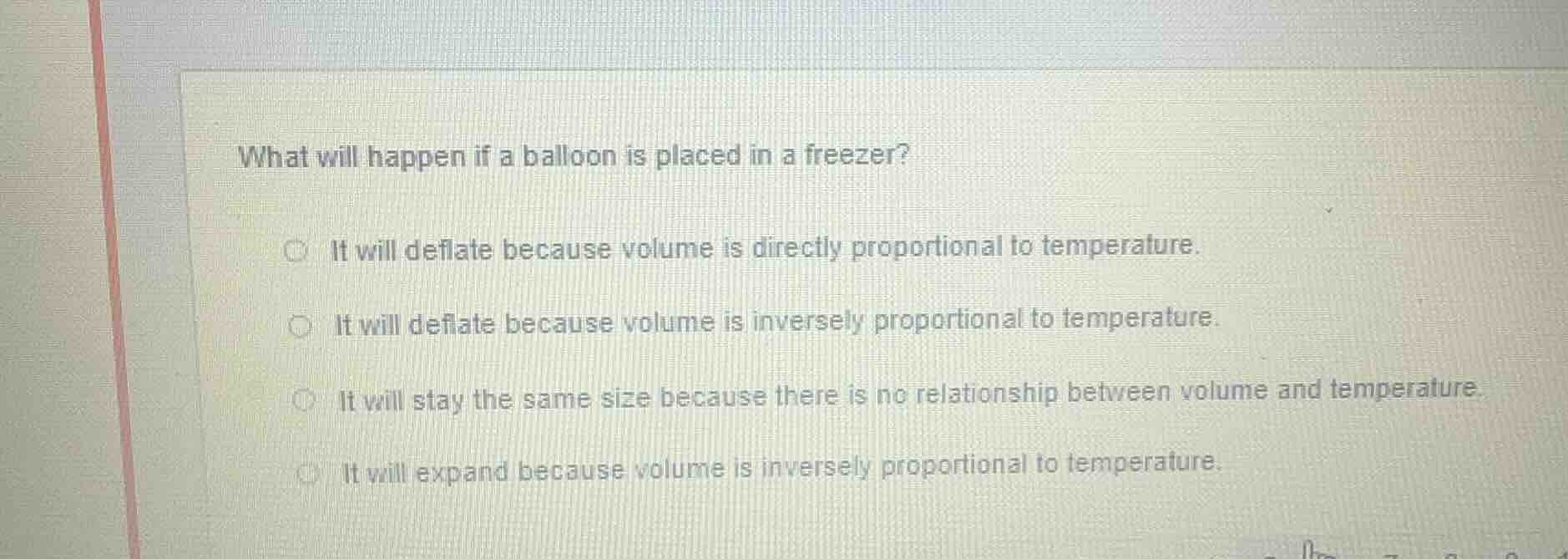
what will happen if a balloon is placed in a freezer? it will deflate because volume is directly proportional to temperature. it will deflate because volume is inversely proportional to temperature. it will stay the same size because there is no relationship between volume and temperature. it will expand because volume is inversely proportional to temperature.
This relies on Charles's Law, which states that for a fixed amount of gas at constant pressure, volume ($V$) and absolute temperature ($T$) are directly proportional, expressed as $\frac{V_1}{T_1}=\frac{V_2}{T_2}$. A freezer lowers the temperature of the gas inside the balloon, so the volume decreases (deflation) as temperature drops.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
It will deflate because volume is directly proportional to temperature.