QUESTION IMAGE
Question
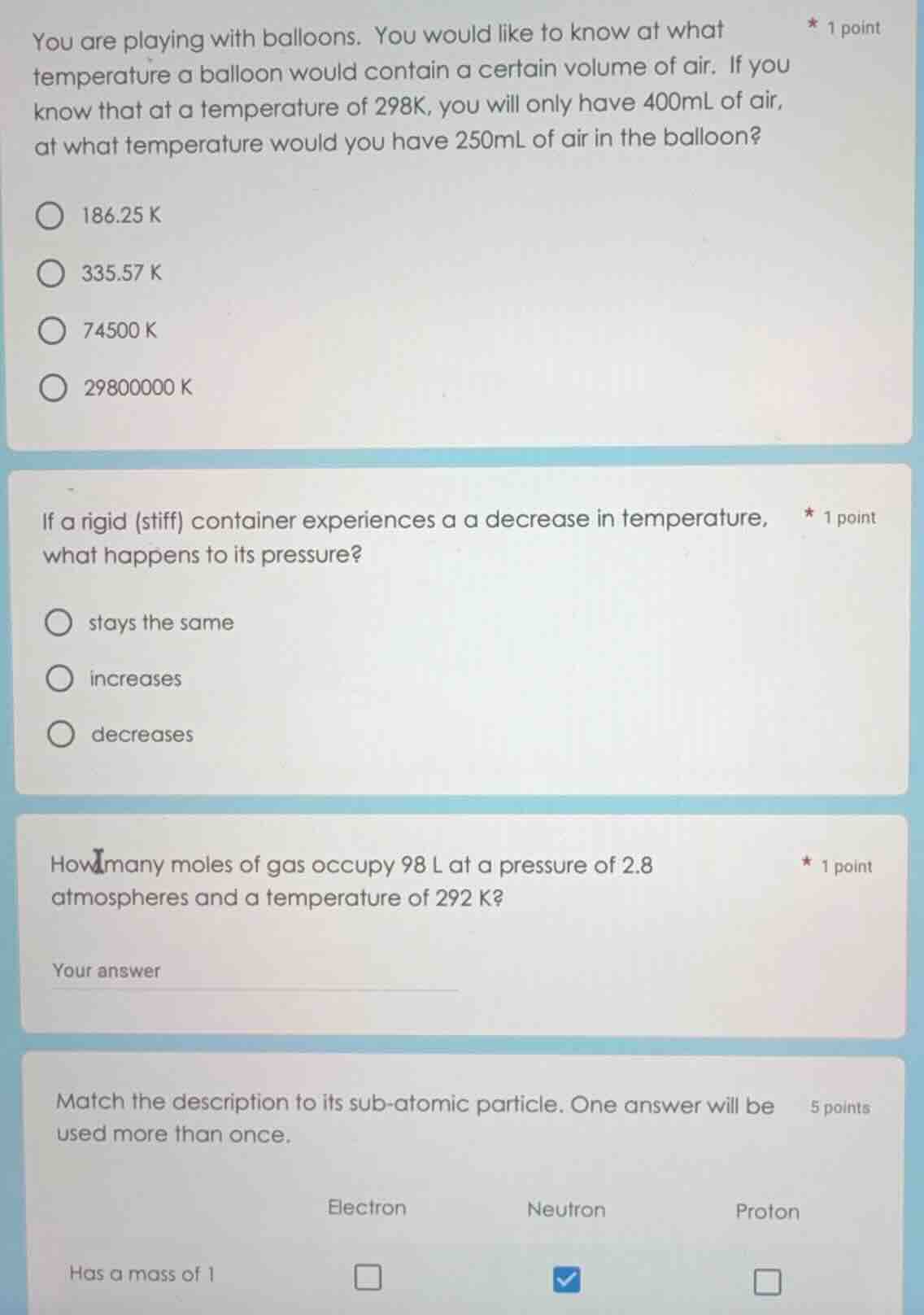
you are playing with balloons. you would like to know at what temperature a balloon would contain a certain volume of air. if you know that at a temperature of 298k, you will only have 400ml of air, at what temperature would you have 250ml of air in the balloon?
186.25 k
335.57 k
74500 k
29800000 k
if a rigid (stiff) container experiences a a decrease in temperature, what happens to its pressure?
stays the same
increases
decreases
how many moles of gas occupy 98 l at a pressure of 2.8 atmospheres and a temperature of 292 k?
your answer
match the description to its sub-atomic particle. one answer will be used more than once.
electron neutron proton
has a mass of 1
First Question (Balloon Temperature)
Step1: Identify Gas Law (Charles's Law)
For a gas (air in balloon) at constant pressure, Charles's Law applies: $\frac{V_1}{T_1}=\frac{V_2}{T_2}$. Here, $V_1 = 400\,\text{mL}$, $T_1 = 298\,\text{K}$, $V_2 = 250\,\text{mL}$, solve for $T_2$.
Step2: Rearrange Formula
$T_2=\frac{V_2\times T_1}{V_1}$
Step3: Substitute Values
$T_2=\frac{250\,\text{mL}\times298\,\text{K}}{400\,\text{mL}}=\frac{74500}{400}=186.25\,\text{K}$
For a rigid (constant volume) container, Gay - Lussac's Law states that pressure and temperature (in Kelvin) are directly proportional ($\frac{P_1}{T_1}=\frac{P_2}{T_2}$). If temperature decreases, pressure decreases (since $P\propto T$ for constant $V$).
Step1: Identify Gas Law (Ideal Gas Law)
Ideal Gas Law: $PV = nRT$, where $P = 2.8\,\text{atm}$, $V = 98\,\text{L}$, $T = 292\,\text{K}$, $R = 0.0821\,\text{L·atm/(mol·K)}$. Solve for $n$.
Step2: Rearrange Formula
$n=\frac{PV}{RT}$
Step3: Substitute Values
$n=\frac{2.8\,\text{atm}\times98\,\text{L}}{0.0821\,\text{L·atm/(mol·K)}\times292\,\text{K}}=\frac{274.4}{23.9732}\approx11.45\,\text{mol}$ (approximate, depending on $R$ precision)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
186.25 K