QUESTION IMAGE
Question
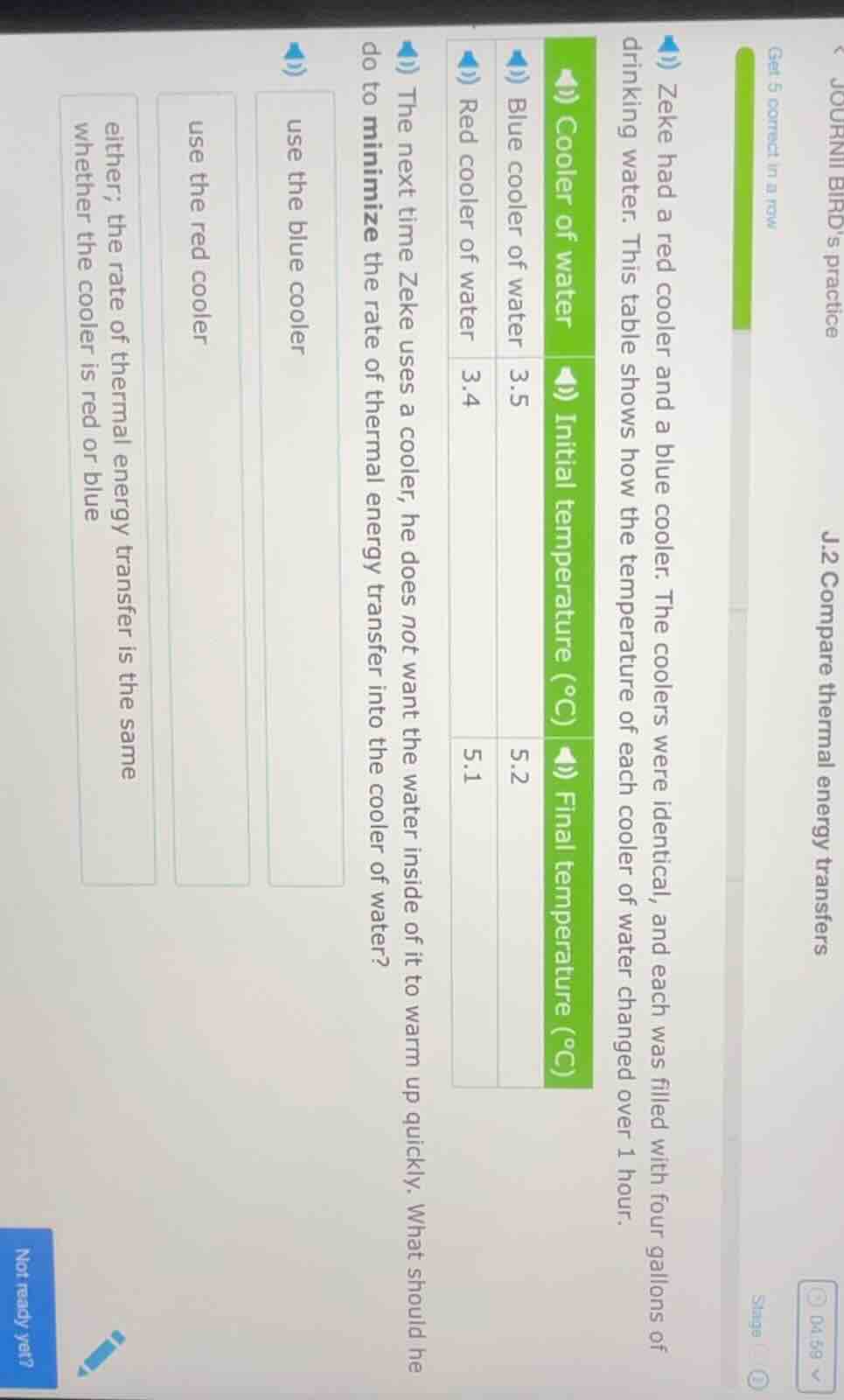
zeke had a red cooler and a blue cooler. the coolers were identical, and each was filled with four gallons of drinking water. this table shows how the temperature of each cooler of water changed over 1 hour. cooler of water initial temperature (°c) final temperature (°c) blue cooler of water 3.5 5.2 red cooler of water 3.4 5.1 the next time zeke uses a cooler, he does not want the water inside of it to warm up quickly. what should he do to minimize the rate of thermal energy transfer into the cooler of water? use the blue cooler use the red cooler either; the rate of thermal energy transfer is the same whether the cooler is red or blue
Step1: Calculate temp change (blue)
$\Delta T_{\text{blue}} = 5.2^\circ\text{C} - 3.5^\circ\text{C} = 1.7^\circ\text{C}$
Step2: Calculate temp change (red)
$\Delta T_{\text{red}} = 5.1^\circ\text{C} - 3.4^\circ\text{C} = 1.7^\circ\text{C}$
Step3: Compare thermal transfer rate
Since mass, time, and $\Delta T$ are equal for both coolers, the thermal energy transfer rate is identical.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
either; the rate of thermal energy transfer is the same whether the cooler is red or blue