QUESTION IMAGE
Question
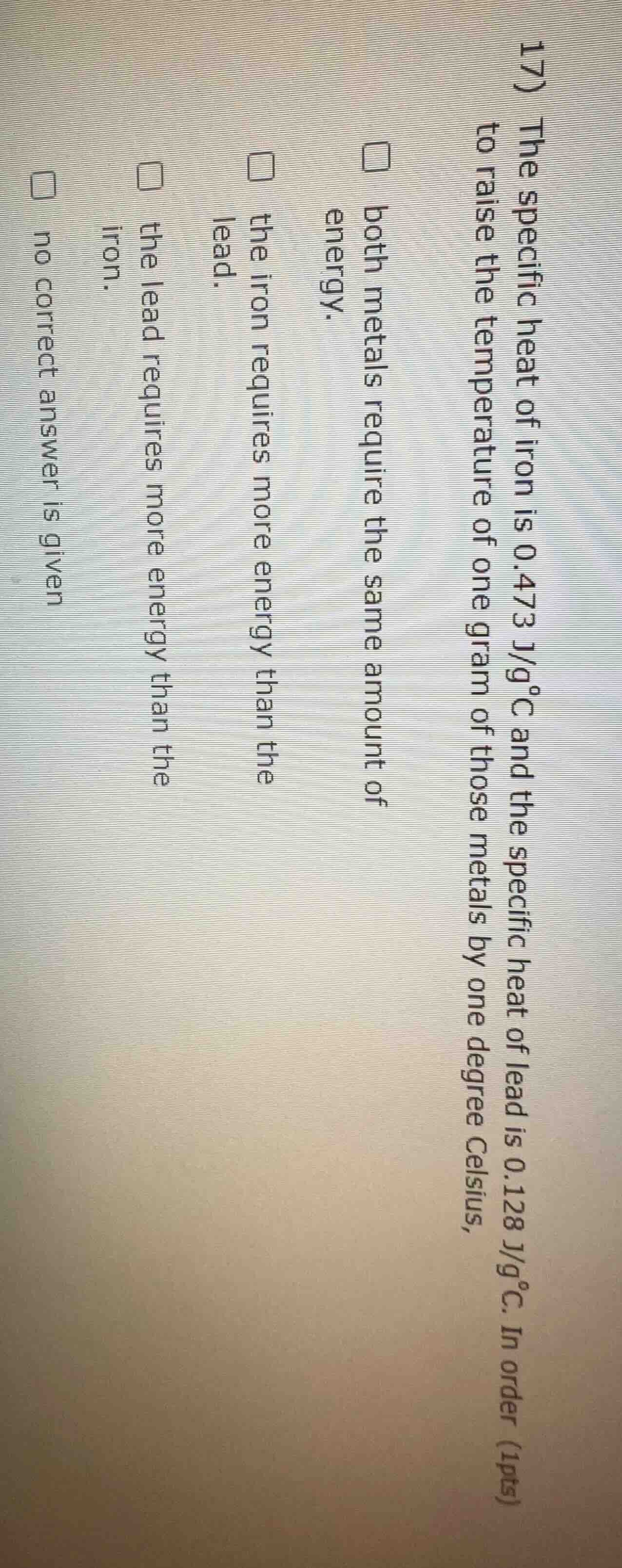
- the specific heat of iron is 0.473 j/g°c and the specific heat of lead is 0.128 j/g°c. in order to raise the temperature of one gram of those metals by one degree celsius, (1pts) both metals require the same amount of energy. the iron requires more energy than the lead. the lead requires more energy than the iron. no correct answer is given
Step1: Recall specific heat formula
The energy required to change temperature is given by $Q = mc\Delta T$, where $m$ = mass, $c$ = specific heat, $\Delta T$ = temperature change.
Step2: Plug in values for iron
For 1g iron, $\Delta T=1^\circ\text{C}$, $c=0.473\ \text{J/g}^\circ\text{C}$:
$Q_{\text{iron}} = 1 \times 0.473 \times 1 = 0.473\ \text{J}$
Step3: Plug in values for lead
For 1g lead, $\Delta T=1^\circ\text{C}$, $c=0.128\ \text{J/g}^\circ\text{C}$:
$Q_{\text{lead}} = 1 \times 0.128 \times 1 = 0.128\ \text{J}$
Step4: Compare the two energies
$0.473\ \text{J} > 0.128\ \text{J}$, so iron requires more energy.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
the iron requires more energy than the lead.