QUESTION IMAGE
Question
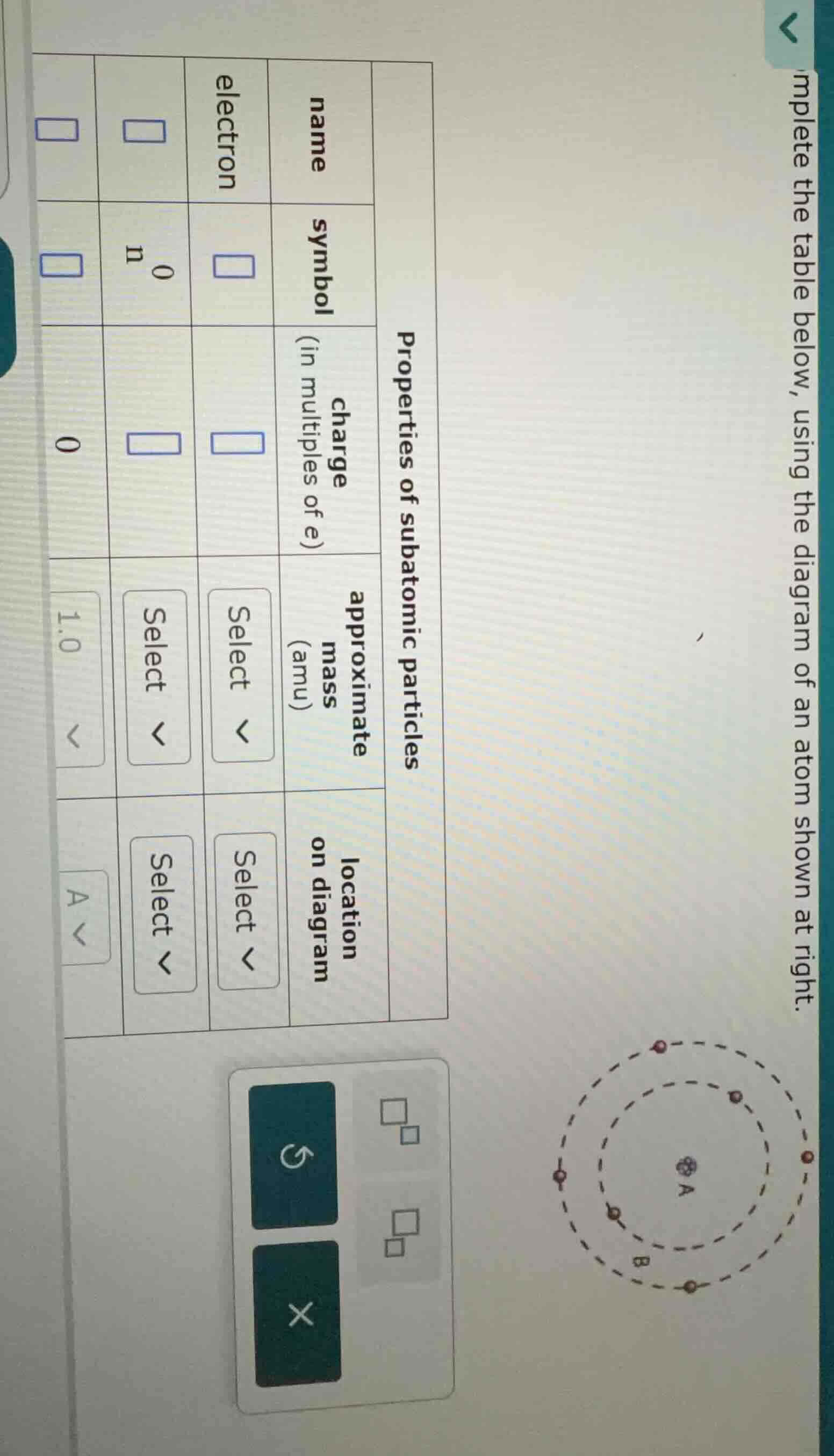
complete the table below, using the diagram of an atom shown at right.
properties of subatomic particles
| name | symbol | charge (in multiples of e) | approximate mass (amu) | location on diagram | |
|---|---|---|---|---|---|
| n | $^0_0$n | ||||
| 0 | 1.0 | a |
To complete the table for subatomic particles (focusing on electron, neutron, and proton, with the given symbol \( \boldsymbol{n} \) for neutron here, but let's address the electron row first, then neutron, and proton context):
For the Electron row:
- Name: Electron
- Symbol: \( e^- \) (or sometimes just \( e \), but with charge indication)
- Charge (in multiples of \( e \)): \( -1 \) (since electron has a negative charge equal in magnitude to proton’s positive charge)
- Approximate mass (amu): \( \approx 0.00054858 \, \text{amu} \) (or often rounded to \( \approx 0 \, \text{amu} \) for simplicity, as it’s much lighter than proton/neutron)
- Location on diagram: In the electron cloud (the outer rings, like the dotted circles labeled B or the outer orbits in the atom diagram)
For the Neutron row (since the symbol given is \( \boldsymbol{n} \)):
- Name: Neutron
- Symbol: \( n \) (matches the given symbol)
- Charge (in multiples of \( e \)): \( 0 \) (neutrons have no charge)
- Approximate mass (amu): \( \approx 1.008665 \, \text{amu} \) (or rounded to \( \approx 1 \, \text{amu} \))
- Location on diagram: In the nucleus (the central “A” region, since neutrons are in the atomic nucleus with protons)
For the Proton row (contextual, as nucleus contains protons/neutrons):
- Name: Proton
- Symbol: \( p^+ \) (or \( p \))
- Charge (in multiples of \( e \)): \( +1 \)
- Approximate mass (amu): \( \approx 1.007276 \, \text{amu} \) (or rounded to \( \approx 1 \, \text{amu} \))
- Location on diagram: In the nucleus (central “A” region)
If the table has columns for electron, neutron, and another particle (like proton), fill each row with these properties. For example, if the first row is “electron”:
- Name: Electron
- Symbol: \( e^- \)
- Charge: \( -1 \)
- Mass: \( \approx 0 \, \text{amu} \) (or \( 0.00055 \, \text{amu} \))
- Location: Electron cloud (outer orbits, e.g., the dotted rings labeled B)
For the row with symbol \( n \) (neutron):
- Name: Neutron
- Symbol: \( n \)
- Charge: \( 0 \)
- Mass: \( \approx 1 \, \text{amu} \) (or \( 1.0087 \, \text{amu} \))
- Location: Nucleus (central “A” region)
Final Table (Example Completion):
| Name | Symbol | Charge (multiples of \( e \)) | Approximate Mass (amu) | Location on Diagram |
|---|---|---|---|---|
| Neutron | \( n \) | \( 0 \) | \( \approx 1 \) | Nucleus (central “A”) |
| Proton | \( p^+ \) | \( +1 \) | \( \approx 1 \) | Nucleus (central “A”) |
Adjust based on the exact table structure (e.g., if only two rows: electron and neutron, or electron and proton). The key is matching each particle’s properties to the columns!
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
To complete the table for subatomic particles (focusing on electron, neutron, and proton, with the given symbol \( \boldsymbol{n} \) for neutron here, but let's address the electron row first, then neutron, and proton context):
For the Electron row:
- Name: Electron
- Symbol: \( e^- \) (or sometimes just \( e \), but with charge indication)
- Charge (in multiples of \( e \)): \( -1 \) (since electron has a negative charge equal in magnitude to proton’s positive charge)
- Approximate mass (amu): \( \approx 0.00054858 \, \text{amu} \) (or often rounded to \( \approx 0 \, \text{amu} \) for simplicity, as it’s much lighter than proton/neutron)
- Location on diagram: In the electron cloud (the outer rings, like the dotted circles labeled B or the outer orbits in the atom diagram)
For the Neutron row (since the symbol given is \( \boldsymbol{n} \)):
- Name: Neutron
- Symbol: \( n \) (matches the given symbol)
- Charge (in multiples of \( e \)): \( 0 \) (neutrons have no charge)
- Approximate mass (amu): \( \approx 1.008665 \, \text{amu} \) (or rounded to \( \approx 1 \, \text{amu} \))
- Location on diagram: In the nucleus (the central “A” region, since neutrons are in the atomic nucleus with protons)
For the Proton row (contextual, as nucleus contains protons/neutrons):
- Name: Proton
- Symbol: \( p^+ \) (or \( p \))
- Charge (in multiples of \( e \)): \( +1 \)
- Approximate mass (amu): \( \approx 1.007276 \, \text{amu} \) (or rounded to \( \approx 1 \, \text{amu} \))
- Location on diagram: In the nucleus (central “A” region)
If the table has columns for electron, neutron, and another particle (like proton), fill each row with these properties. For example, if the first row is “electron”:
- Name: Electron
- Symbol: \( e^- \)
- Charge: \( -1 \)
- Mass: \( \approx 0 \, \text{amu} \) (or \( 0.00055 \, \text{amu} \))
- Location: Electron cloud (outer orbits, e.g., the dotted rings labeled B)
For the row with symbol \( n \) (neutron):
- Name: Neutron
- Symbol: \( n \)
- Charge: \( 0 \)
- Mass: \( \approx 1 \, \text{amu} \) (or \( 1.0087 \, \text{amu} \))
- Location: Nucleus (central “A” region)
Final Table (Example Completion):
| Name | Symbol | Charge (multiples of \( e \)) | Approximate Mass (amu) | Location on Diagram |
|---|---|---|---|---|
| Neutron | \( n \) | \( 0 \) | \( \approx 1 \) | Nucleus (central “A”) |
| Proton | \( p^+ \) | \( +1 \) | \( \approx 1 \) | Nucleus (central “A”) |
Adjust based on the exact table structure (e.g., if only two rows: electron and neutron, or electron and proton). The key is matching each particle’s properties to the columns!