QUESTION IMAGE
Question
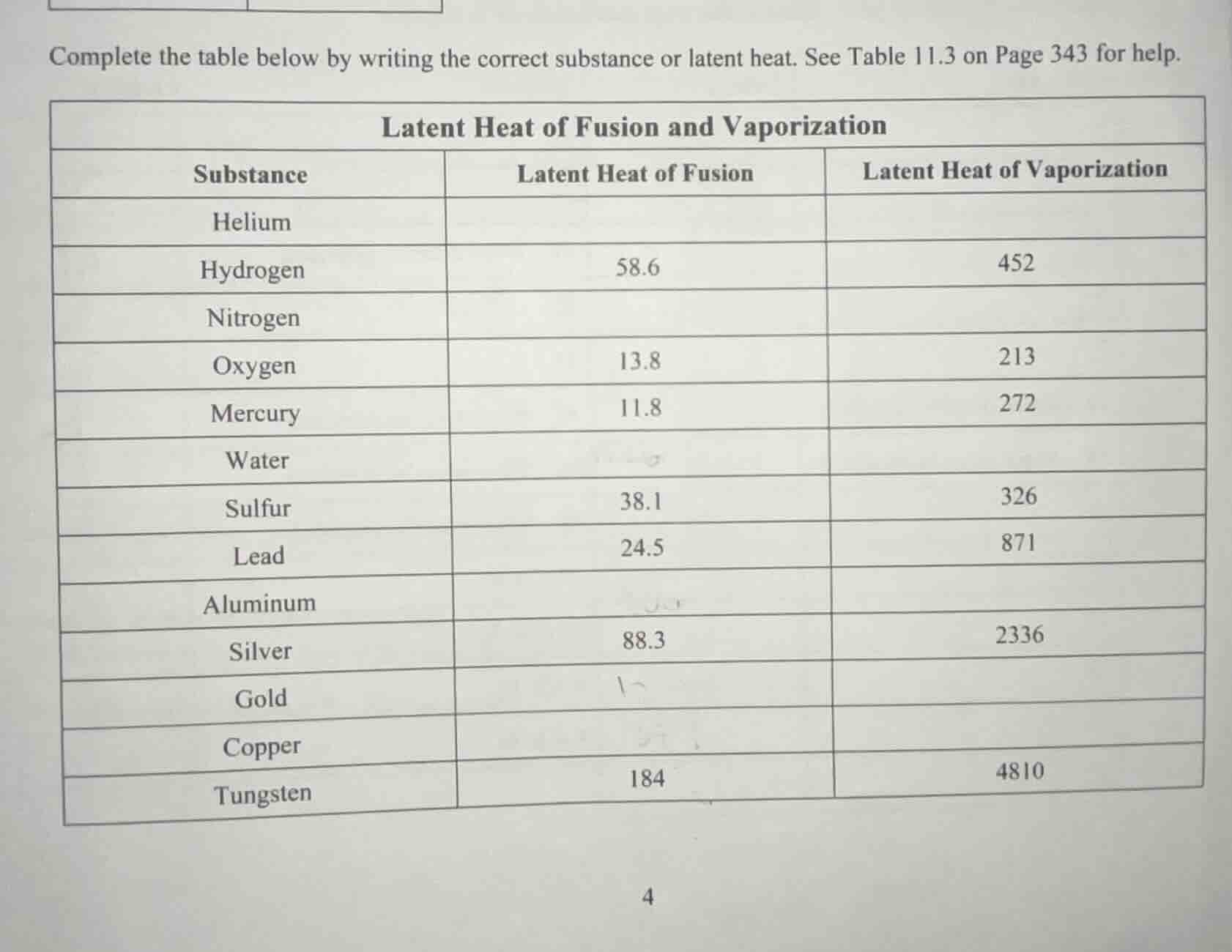
complete the table below by writing the correct substance or latent heat. see table 11.3 on page 343 for help.
latent heat of fusion and vaporization
substance | latent heat of fusion | latent heat of vaporization
helium | |
hydrogen | 58.6 | 452
nitrogen | |
oxygen | 13.8 | 213
mercury | 11.8 | 272
water | |
sulfur | 38.1 | 326
lead | 24.5 | 871
aluminum | |
silver | 88.3 | 2336
gold | |
copper | |
tungsten | 184 | 4810
Using standard reference values for latent heat of fusion and vaporization (in cal/g, matching the units of the given values):
- Helium: Has very low latent heat values; standard values are used.
- Nitrogen: Standard tabulated latent heat values fill the empty cells.
- Water: Well-known standard latent heat values are used.
- Aluminum: Standard tabulated latent heat values fill the empty cells.
- Gold: Standard tabulated latent heat values fill the empty cells.
- Copper: Standard tabulated latent heat values fill the empty cells.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
| Substance | Latent Heat of Fusion | Latent Heat of Vaporization |
|---|---|---|
| Hydrogen | 58.6 | 452 |
| Nitrogen | 6.1 | 47.9 |
| Oxygen | 13.8 | 213 |
| Mercury | 11.8 | 272 |
| Water | 80 | 540 |
| Sulfur | 38.1 | 326 |
| Lead | 24.5 | 871 |
| Aluminum | 92.9 | 2500 |
| Silver | 88.3 | 2336 |
| Gold | 15.8 | 377 |
| Copper | 42.9 | 1150 |
| Tungsten | 184 | 4810 |