QUESTION IMAGE
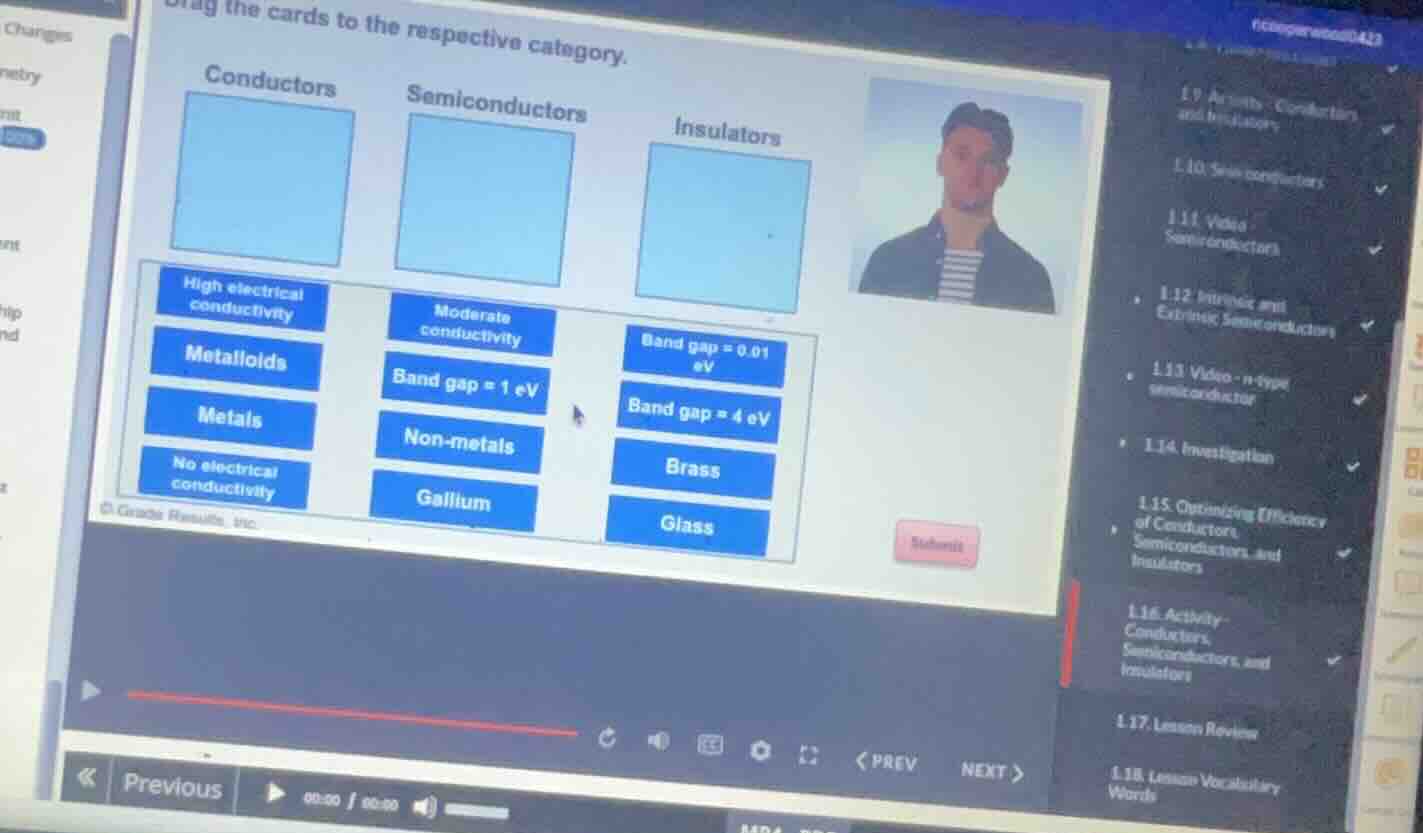
Question
drag the cards to the respective category.
conductors semiconductors insulators
high electrical conductivity metalloids metals no electrical conductivity moderate conductivity band gap = 1 ev non - metals gallium band gap = 0.01 ev band gap = 4 ev brass glass
Brief Explanations
To solve this, we use the properties of conductors, semiconductors, and insulators:
Conductors:
- Have high electrical conductivity (electrons move freely).
- Include Metals (e.g., copper, iron) and alloys like Brass (metal - based, conducts well).
- “Band gap = 0.01 eV” is very small (almost no gap), so electrons can easily move → conductor.
Semiconductors:
- Have moderate conductivity (between conductors and insulators).
- Include Metalloids (e.g., silicon, germanium) and materials with “Band gap = 1 eV” (typical for semiconductors, e.g., silicon has ~1.1 eV).
- “Gallium” is a metalloid - like element used in semiconductors (e.g., gallium arsenide).
Insulators:
- Have no electrical conductivity (electrons are tightly bound).
- Have large band gaps (e.g., “Band gap = 4 eV” for materials like glass, which is an insulator).
- “Non - metals” like sulfur, carbon (in diamond form) are insulators.
Categorization:
- Conductors: High electrical conductivity, Metals, Brass, Band gap = 0.01 eV
- Semiconductors: Moderate conductivity, Metalloids, Band gap = 1 eV, Gallium
- Insulators: No electrical conductivity, Non - metals, Band gap = 4 eV, Glass
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- Conductors: High electrical conductivity, Metals, Brass, Band gap = 0.01 eV
- Semiconductors: Moderate conductivity, Metalloids, Band gap = 1 eV, Gallium
- Insulators: No electrical conductivity, Non - metals, Band gap = 4 eV, Glass