QUESTION IMAGE
Question
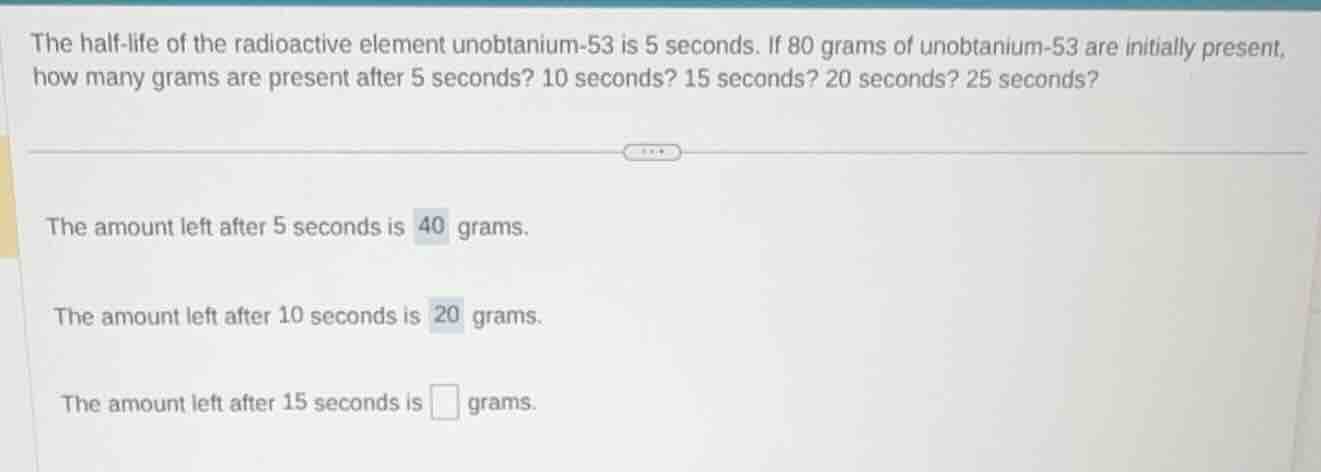
the half-life of the radioactive element unobtanium-53 is 5 seconds. if 80 grams of unobtanium-53 are initially present, how many grams are present after 5 seconds? 10 seconds? 15 seconds? 20 seconds? 25 seconds?
the amount left after 5 seconds is 40 grams.
the amount left after 10 seconds is 20 grams.
the amount left after 15 seconds is \boxed{} grams.
Step1: Understand half - life concept
The half - life of a radioactive substance is the time it takes for half of the substance to decay. So, after each half - life period, the amount of the substance is multiplied by $\frac{1}{2}$.
Step2: Calculate amount after 15 seconds
We know that the initial amount $A_0 = 80$ grams and the half - life $t_{1/2}=5$ seconds.
After 5 seconds (1 half - life), the amount $A_1=A_0\times\frac{1}{2}=80\times\frac{1}{2} = 40$ grams.
After 10 seconds (2 half - lives), the amount $A_2 = A_1\times\frac{1}{2}=40\times\frac{1}{2}=20$ grams.
After 15 seconds, we have passed 3 half - lives (since $15\div5 = 3$). So we take the amount after 10 seconds and multiply by $\frac{1}{2}$ again.
$A_3=A_2\times\frac{1}{2}=20\times\frac{1}{2}=10$ grams.
Step3: Calculate amount after 20 seconds
After 20 seconds, we have passed 4 half - lives (since $20\div5 = 4$). We take the amount after 15 seconds and multiply by $\frac{1}{2}$.
$A_4=A_3\times\frac{1}{2}=10\times\frac{1}{2} = 5$ grams.
Step4: Calculate amount after 25 seconds
After 25 seconds, we have passed 5 half - lives (since $25\div5=5$). We take the amount after 20 seconds and multiply by $\frac{1}{2}$.
$A_5=A_4\times\frac{1}{2}=5\times\frac{1}{2}=\frac{5}{2} = 2.5$ grams.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- After 15 seconds: 10 grams.
- After 20 seconds: 5 grams.
- After 25 seconds: 2.5 grams.