QUESTION IMAGE
Question
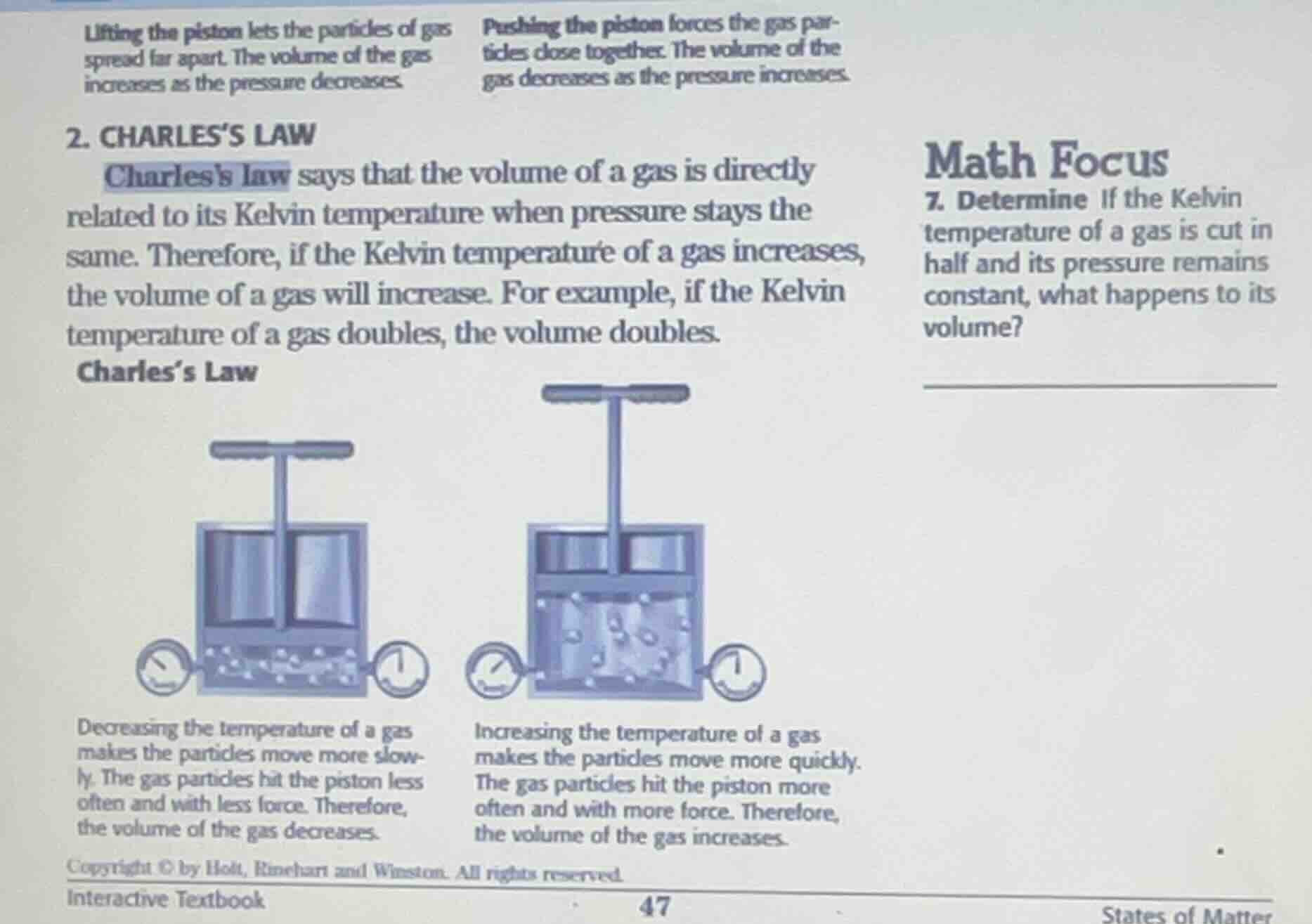
lifting the piston lets the particles of gas spread far apart. the volume of the gas increases as the pressure decreases. pushing the piston forces the gas particles close together. the volume of the gas decreases as the pressure increases. 2. charles’s law charless law says that the volume of a gas is directly related to its kelvin temperature when pressure stays the same. therefore, if the kelvin temperature of a gas increases, the volume of a gas will increase. for example, if the kelvin temperature of a gas doubles, the volume doubles. charles’s law decreasing the temperature of a gas makes the particles move more slowly. the gas particles hit the piston less often and with less force. therefore, the volume of the gas decreases. increasing the temperature of a gas makes the particles move more quickly. the gas particles hit the piston more often and with more force. therefore, the volume of the gas increases. copyright © by holt, rinehart and winston. all rights reserved. interactive textbook math focus 7. determine if the kelvin temperature of a gas is cut in half and its pressure remains constant, what happens to its volume?
Step1: State Charles's Law formula
Charles's Law (constant pressure) is $\frac{V_1}{T_1} = \frac{V_2}{T_2}$, where $V_1$ = initial volume, $T_1$ = initial Kelvin temperature, $V_2$ = final volume, $T_2$ = final Kelvin temperature.
Step2: Define the temperature change
Given $T_2 = \frac{1}{2}T_1$. Rearrange the law to solve for $V_2$:
$V_2 = V_1 \times \frac{T_2}{T_1}$
Step3: Substitute $T_2$ into the formula
Substitute $T_2 = \frac{1}{2}T_1$:
$V_2 = V_1 \times \frac{\frac{1}{2}T_1}{T_1}$
Step4: Simplify to find $V_2$
Cancel $T_1$ in numerator and denominator:
$V_2 = \frac{1}{2}V_1$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
The volume of the gas is cut in half (decreases to one-half of its original volume).