QUESTION IMAGE
Question
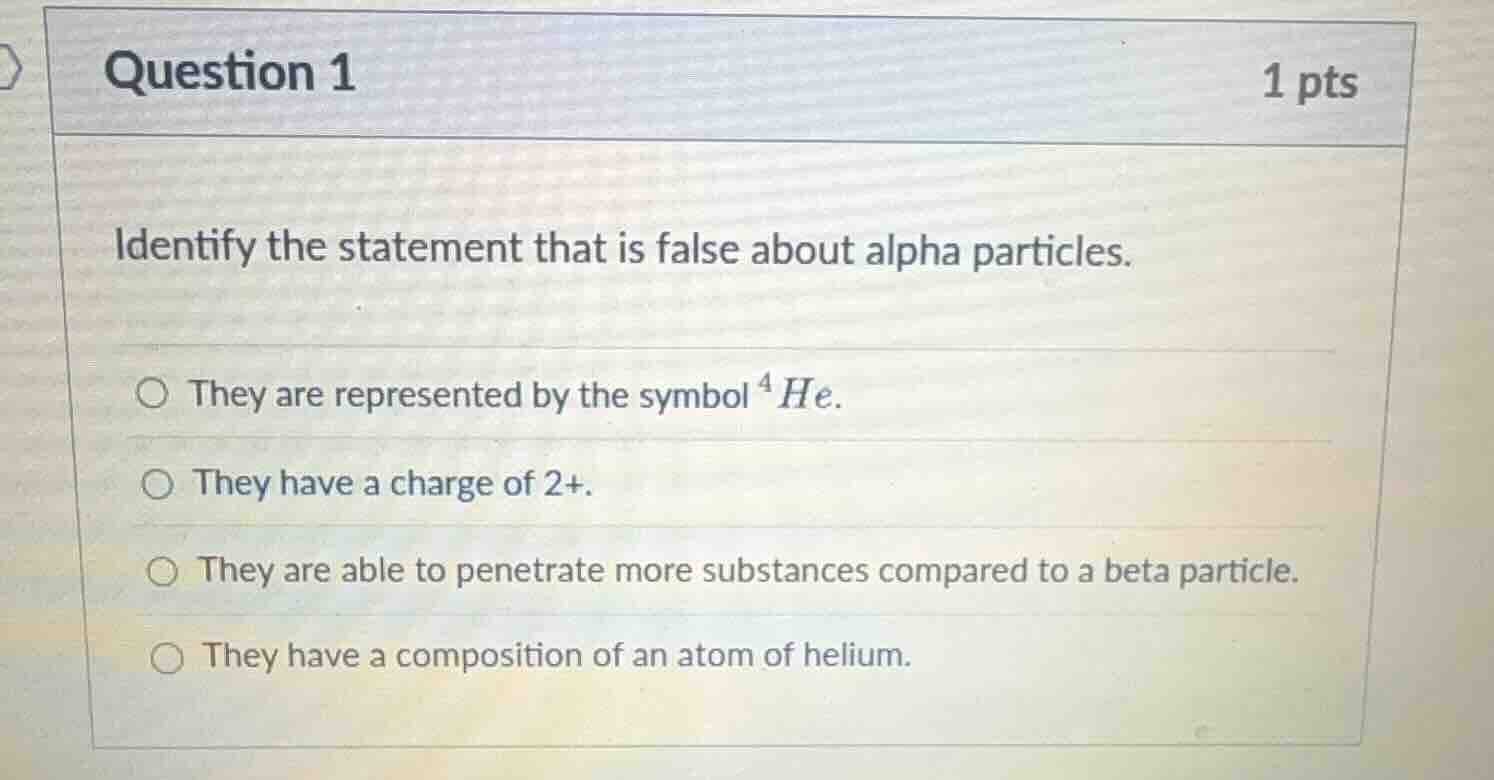
question 1
1 pts
identify the statement that is false about alpha particles.
they are represented by the symbol $^4he$.
they have a charge of 2+.
they are able to penetrate more substances compared to a beta particle.
they have a composition of an atom of helium.
Brief Explanations
- Recall properties of alpha particles:
- Alpha particles are \( _2^4He^{2+} \) (or represented as \( ^4He \) for the particle, with charge \( 2+ \)), so the first two statements are true.
- Alpha particles have low penetration power (due to large mass and charge), while beta particles (smaller, less charge) penetrate more. So "They are able to penetrate more substances compared to a beta particle" is false.
- Alpha particles consist of 2 protons and 2 neutrons, same as a helium nucleus (atom of helium has electrons, but the particle is like the nucleus, so the composition statement is true in context of the particle's structure).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
They are able to penetrate more substances compared to a beta particle.