QUESTION IMAGE
Question
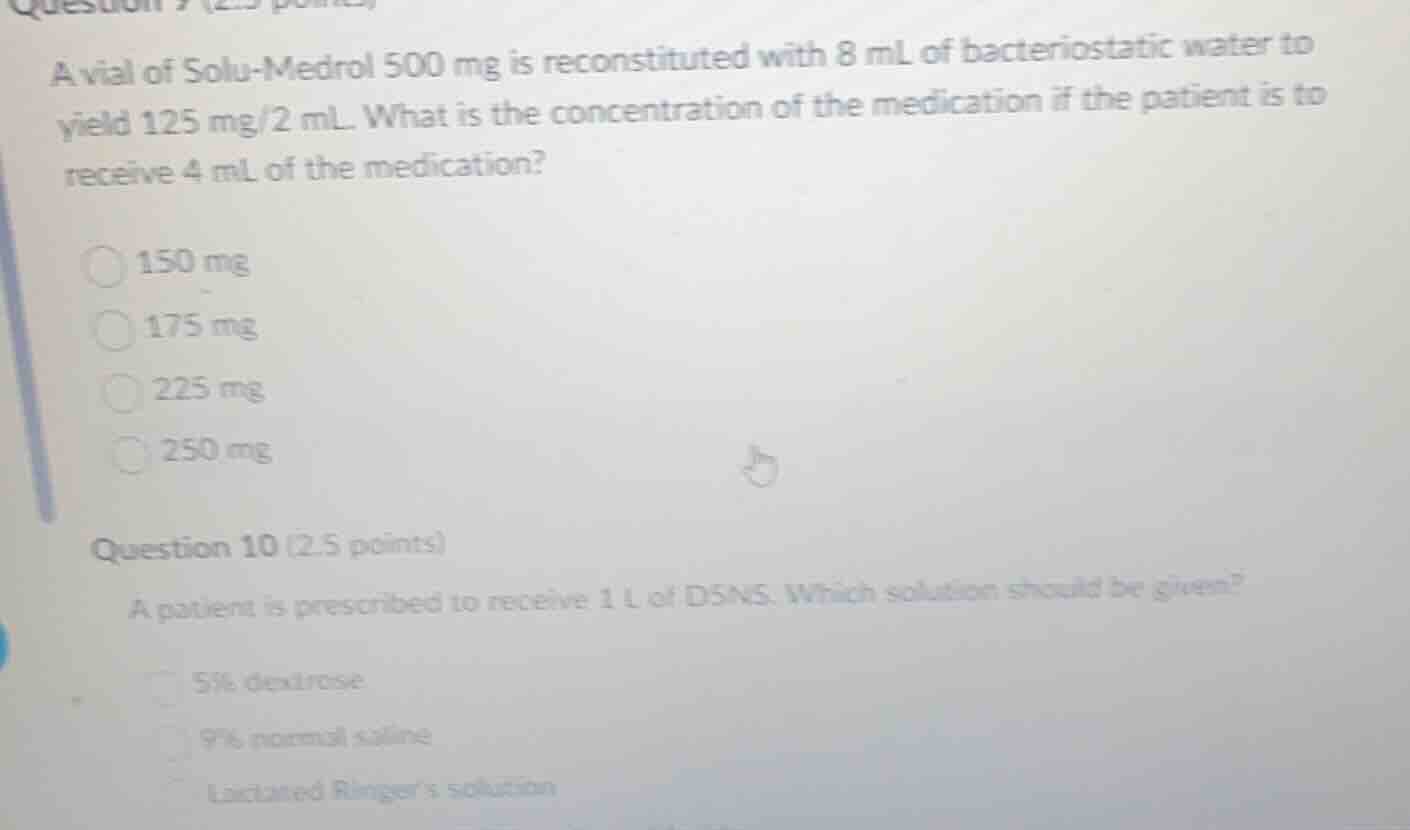
a vial of solu - medrol 500 mg is reconstituted with 8 ml of bacteriostatic water to yield 125 mg/2 ml. what is the concentration of the medication if the patient is to receive 4 ml of the medication?
150 mg
175 mg
225 mg
250 mg
question 10 (2.5 points)
a patient is prescribed to receive 1 l of d5ns. which solution should be given?
5% dextrose
9% normal saline
lactated ringer’s solution
Question 9
Step1: Determine the concentration per mL
The concentration is given as 125 mg/2 mL. To find the concentration per mL, we divide 125 mg by 2 mL.
$Concentration\ per\ mL = \frac{125\ mg}{2\ mL} = 62.5\ mg/mL$
Step2: Calculate the amount in 4 mL
Now, to find the amount in 4 mL, we multiply the concentration per mL by 4 mL.
$Amount\ in\ 4\ mL = 62.5\ mg/mL\times4\ mL = 250\ mg$
D5NS stands for 5% dextrose in normal saline. So the solution that should be given is 5% dextrose (along with normal saline, but among the options, 5% dextrose is the relevant component here).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
250 mg