QUESTION IMAGE
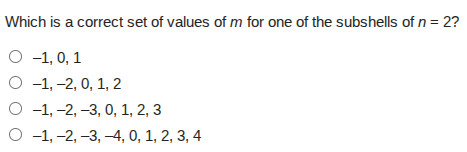
Question
which is a correct set of values of m for one of the subshells of n = 2?
−1, 0, 1
−1, −2, 0, 1, 2
−1, −2, −3, 0, 1, 2, 3
−1, −2, −3, −4, 0, 1, 2, 3, 4
(since $l = 0,1,...,n-1$).
Step2: Find m for l=1
The magnetic quantum number $m$ ranges from $-l$ to $+l$ in integer steps. For $l=1$, $m = -1, 0, 1$.
Step3: Eliminate invalid options
Options with values outside $-1$ to $1$ (for $l=1$) or mismatched ranges for valid $l$ values are incorrect. Only the first option matches a valid set of $m$ values for a subshell of $n=2$.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
(since $l = 0,1,...,n-1$).
Step2: Find m for l=1
The magnetic quantum number $m$ ranges from $-l$ to $+l$ in integer steps. For $l=1$, $m = -1, 0, 1$.
Step3: Eliminate invalid options
Options with values outside $-1$ to $1$ (for $l=1$) or mismatched ranges for valid $l$ values are incorrect. Only the first option matches a valid set of $m$ values for a subshell of $n=2$.